Arizona Urology Specialists, Tucson, Arizona
Case Report
A 54-year-old man presented to the urology department for a prostate-specific antigen (PSA) level greater than 1000 µg/L (upper limit of the laboratory assay). The patient’s medical history included type 2 diabetes, hyperlipidemia, and a family history (father) of prostate cancer. A digital rectal exam revealed a firm, nodular prostate. A transrectal ultrasound–guided prostate biopsy was performed, and pathology confirmed adenocarcinoma of the prostate, with the highest Gleason score of 10 (5 + 5) in 5 cores and Gleason score 9 (5 + 4) in 6 cores. The clinical staging was T4bN0M1. The patient’s prostate risk group was very high risk according to National Comprehensive Cancer Network guidelines. Germline genetic testing was negative for targetable genes.
A computed tomography scan of the patient’s abdomen and pelvis with intravenous contrast performed at the time revealed high-volume metastatic osseous disease. A bone scan was then performed that showed widespread radiotracer-avid osseous metastatic disease. A prostate-specific membrane antigen positron emission tomography (PSMA PET) scan was not obtained as part of the initial staging workup.
Androgen-deprivation therapy was initiated with degarelix, and the patient has remained on primary hormone therapy, with leuprolide injections every 6 months. Because of the patient’s high-volume metastatic disease and high Gleason score, he was also started on darolutamide through the urology department and saw the oncology department to complete 6 cycles of docetaxel chemotherapy. He was also started on zoledronic acid intravenous infusion for bone health. Per National Comprehensive Cancer Network guidelines, no prostate radiation was performed.
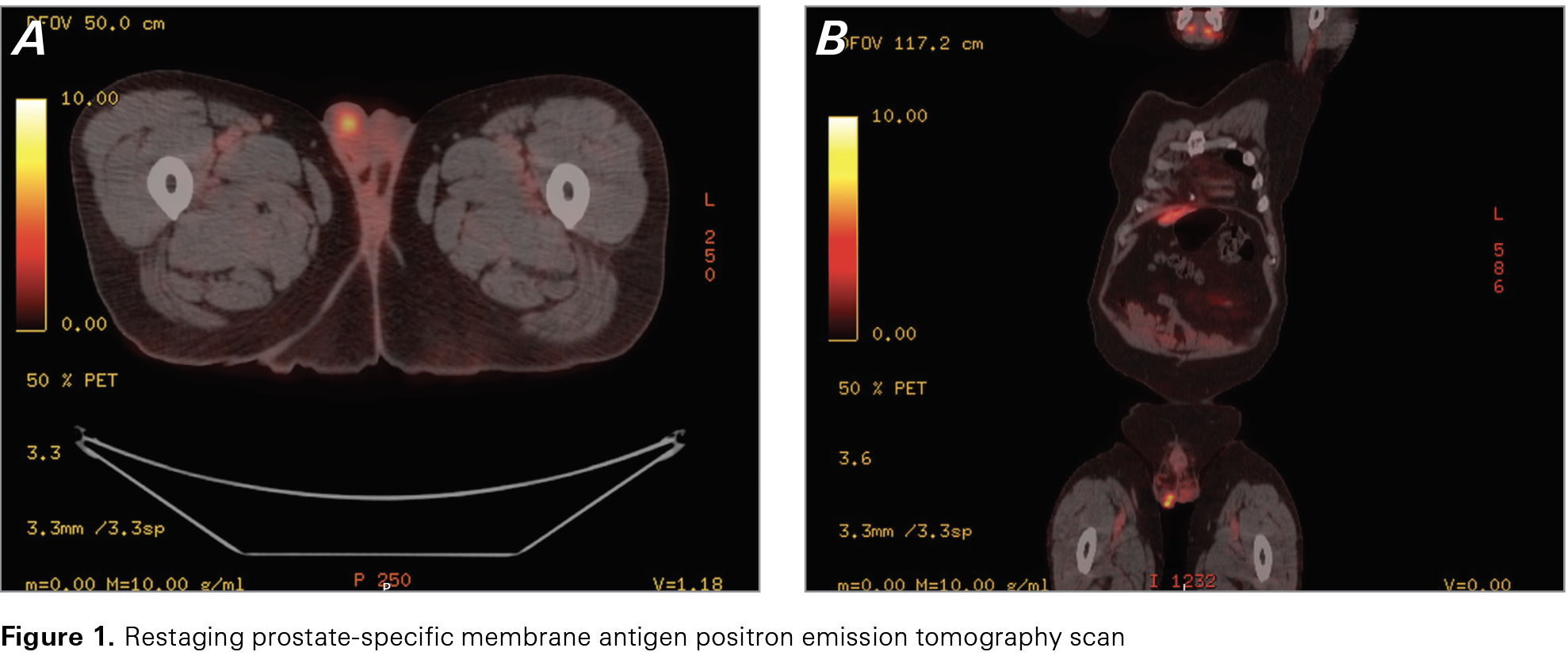
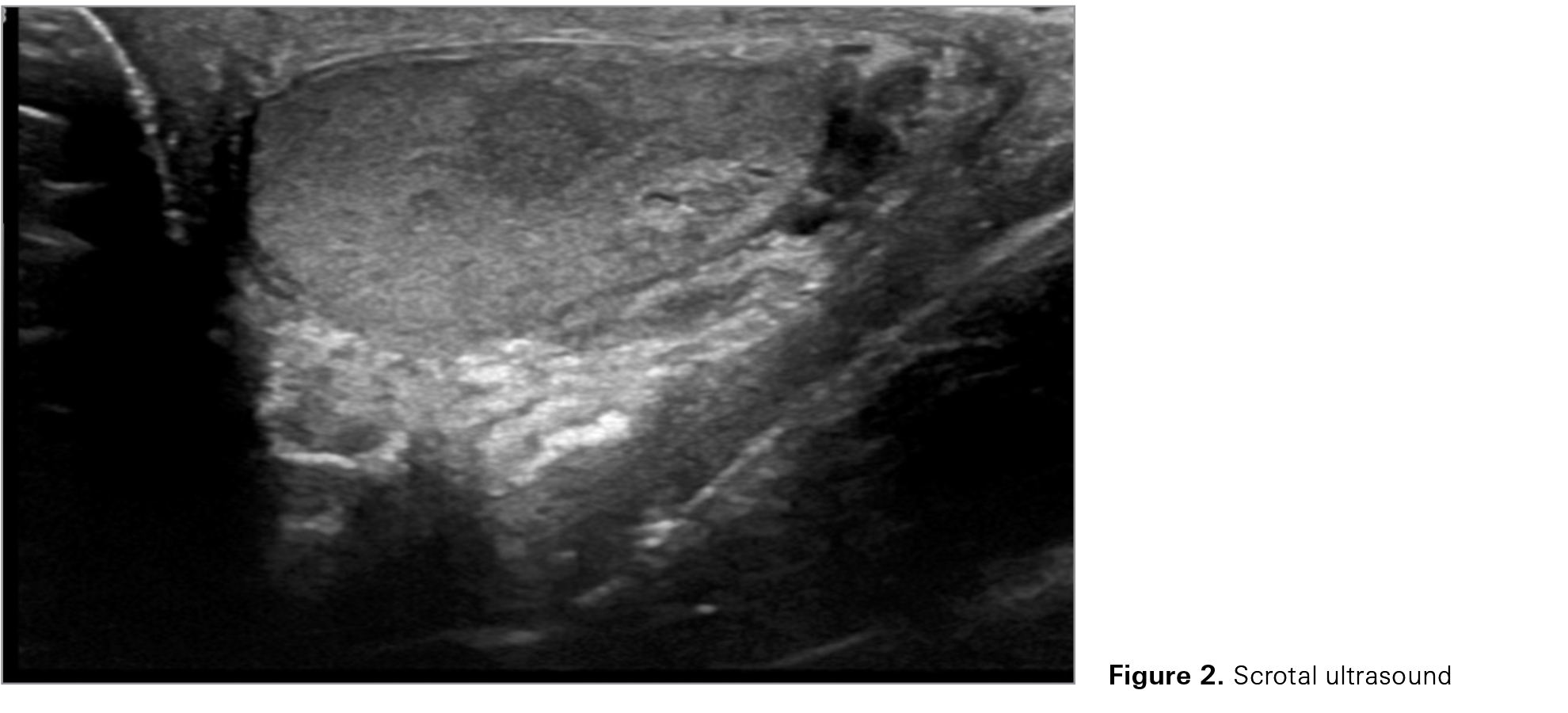
After chemotherapy was finished, the patient’s PSA responded favorably, dropping as low as 0.38 µg/L at 13 months after the patient started treatment. However, 3 months later, biochemical progression was seen when the patient’s PSA level rose to 0.72 µg/L, despite leuprolide and darolutamide. This increase prompted a restaging PSMA PET scan (Figure 1). The scan showed PSMA avidity only in the prostate and in the right testis, suspected to be either prostate cancer metastasis or primary testicular cancer. No other sites of metastasis were seen on the PSMA PET scan. On physical exam, there was no palpable testicular mass, and the patient was asymptomatic. A follow-up scrotal ultrasound confirmed indeterminate, multifocal, rounded, hypoechoic intratesticular nodules or masses in the right testis (Figure 2). Tumor markers α-fetoprotein, lactate dehydrogenase, and b-human chorionic gonadotropin all were normal.
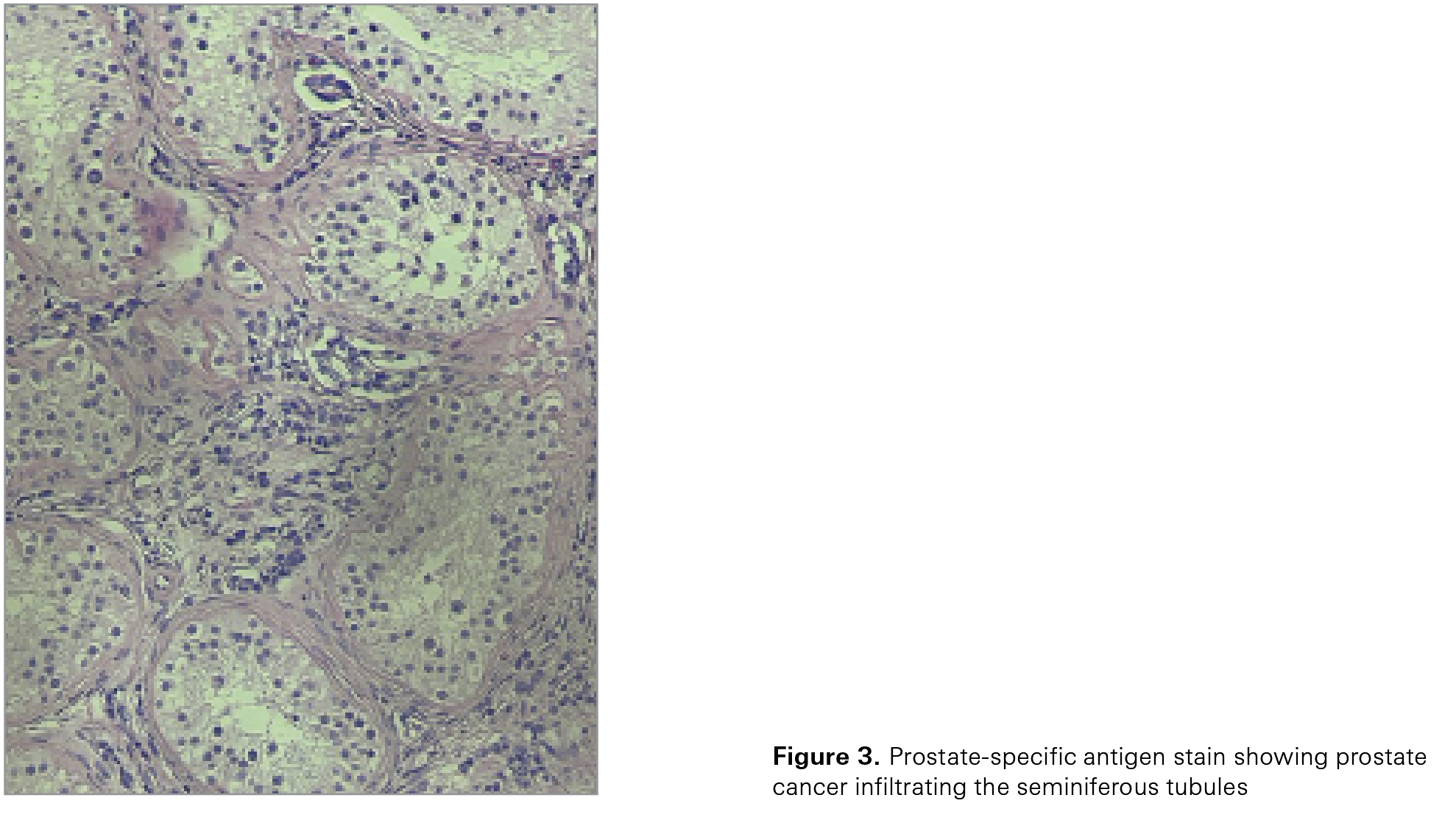
The patient then underwent a radical right orchiectomy. Gross pathologic findings revealed multifocal tan lesions ranging from 0.3 to 0.8 cm in size. These lesions did not grossly invade the tunica albuginea. Microscopic findings, including PSA stain, confirmed metastatic carcinoma of the prostate with negative margins. Standard hematoxylin-eosin stain showed tumor infiltrating between seminiferous tubules (Figure 3). After the orchiectomy, the patient’s PSA level dropped to 0.2 µg/L. The patient then consulted with a radiation oncologist, who offered the option of radiation therapy to the prostate. The patient wanted to be as aggressive as possible and underwent radiation to the prostate (20 fractions of 2.75 Gy for a total dose of 55 Gy). His most recent PSA level was 0.26 µg/L. The patient’s metastatic castration-sensitive prostate cancer appears to be stable on darolutamide and leuprolide, which the urology and oncology departments are planning to continue.
Discussion
In patients with metastatic prostate cancer, the most common sites of metastasis are bone (84%), distant lymph nodes (10.6%), liver (10.2%), and thorax (9.2%).1 Metastasis of prostate cancer to the testicles is rare. Bubendorf et al2 found that in a cohort of 1589 patients with prostate cancer, 35% had hematogenous metastases. Of those patients
who had metastases, the most common sites of involvement were bone (90%), lung (46%), and liver (25%). Testicular metastases were found in 0.5% of patients (n = 3). Overall, with the exception of leukemia and lymphoma infiltration, reported incidences of secondary neoplasms of the testis are low, ranging from 0.02% to 2.5%.3 However, the most common primary site of testicular metastases is the prostate, at approximately 15%.3 The first case of prostate cancer with testicular metastasis was reported by Semans in 1938.4
Secondary metastases to the testes are typically observed in patients who are in their sixth or seventh decade of life, and such metastases often signify advanced disease.5 This patient, who is in his fifth decade of life, represents an unusual case. Given the presence of high-volume metastases at diagnosis, it is plausible that the testicular metastasis was present at diagnosis but unrecognized at that time. Because primary testicular germ cell neoplasms usually occur in younger men, testicular masses seen on imaging in older men should raise suspicion for possible metastases.
Prostate-specific membrane antigen is a type II transmembrane glycoprotein that was discovered in 1987 in metastatic prostate cancer cell lines.6 Expression of PSMA occurs in normal epithelial prostate cells but is markedly upregulated in prostate cancer cells, correlative to the aggressiveness of the disease. Expression typically becomes more intense in cases with high Gleason score and in poorly differentiated, castration-resistant, and metastatic tumors.6
The advent of PSMA PET scanning coupled with serial monitoring of PSA levels has substantially improved the early detection of disease recurrence and metastatic spread. Currently, PSA is considered the most accurate biomarker of disease recurrence and progression.7 In concurrence with this case, the patient’s PSA level fell to 0.38 µg/L from more than 1000 µg/L after the patient finished docetaxel chemotherapy. Then, it rose again to 0.72 µg/L, indicating disease progression.
Because prostatic adenocarcinoma metastasizing to the testes is often a sign of advanced disease, other sites of metastasis in this patient’s PSMA PET scan were expected, especially given his high volume of metastasis at diagnosis. However, his scan revealed PSMA avidity only in the prostate and right testis, with no other sites of metastasis detected. In addition, although testicular swelling (with or without pain) is a recognized symptom of testicular metastasis,3 the patient in this case was asymptomatic, with the mass being nonpalpable. The testicular lesion was detected only because of the restaging PSMA PET scan.
Although radiation to the prostate is not within National Comprehensive Cancer Network guidelines in this situation, the patient received radiation to the prostate after his orchiectomy because he wanted to be as aggressive as possible. This case was presented at our local tumor board, and radiation to the prostate was determined to be reasonable, given that the patient’s PSMA PET scan showed no current metastatic sites. The patient’s young age—54 years—was a factor in the discussion and the decision to employ radiation therapy, as well.
In patients with prostate cancer metastatic to the testes, prognosis is often not favorable because this metastasis signifies advanced disease. Lu et al8 conducted a study that found a mean survival after orchiectomy of 12.8 months. Yet our patient appears to be temporarily stable on his current therapy, with his most recent PSA level at 0.26 µg/L. Further studies are needed to assess mortality rates of patients with prostate cancer who develop metastases to the testes.
Conclusion
Because prostate cancer metastases to the testes are rare, this case demonstrates the importance of close serial PSA monitoring and the value of PSMA PET scanning in prostate cancer treatment. These scans substantially increase diagnostic accuracy and improve treatment decisions. Although the prognosis is often not favorable for patients with prostate cancer metastatic to the testes, this patient’s PSA has responded favorably after his right orchiectomy, and his prostate cancer appears to be responding to darolutamide and leuprolide. Continued monitoring and adaptation of therapy will remain essential.
References
1. Gandaglia G, Abdollah F, Schiffmann J, et al. Distribution of metastatic sites in patients with prostate cancer: a population-based analysis. Prostate. 2014;74(2):210-216. doi:10.1002/pros.22742
2. Bubendorf L, Schöpfer A, Wagner U, et al. Metastatic patterns of prostate cancer: an autopsy study of 1,589 patients. Hum Pathol. 2000;31:578-583. doi:10.1053/hp.2000.6698
3. Kusaka A, Koie T, Yamamoto H, et al. Testicular metastasis of prostate cancer: a case report. Case Rep Oncol. 2014;7(3):643-647. doi:10.1159/000367779
4. Semans JH. Carcinoma of the prostate with metastasis in the testis. J Urol. 1938;40(4):524-529. doi:10.1016/S0022-5347(17)71794-6
5. Turk A, Graff JM, Memo M. Case report of metastatic prostate cancer to testicles: an ominous sign of advanced disease. Urol Case Rep. 2019;26:100935. doi:10.1016/j.eucr.2019.100935
6. de Galiza Barbosa F, Queiroz MA, Nunes RF, et al. Nonprostatic diseases on PSMA PET imaging: a spectrum of benign and malignant findings. Cancer Imaging. 2020;20(1):23. doi:10.1186/s40644-020-00300-7
7. Chen Y, Lin Y, Nie P, et al. Associations of prostate-specific antigen, prostate carcinoma tissue Gleason score, and androgen receptor expression with bone metastasis in patients with prostate carcinoma. Med Sci Monit. 2017;23:1768-1774. doi:10.12659/msm.900977
8. Lu L, Kuo J, Lin ATL, et al. Metastatic tumors involving the testes. J Urol ROC. 2000;11:12-16.
Article Information
Published: 3/31/2025.
Conflict of Interest Disclosures: None.
Funding/Support: None.
Author Contributions: Both authors contributed equally to this article.
Data Availability Statement: No new data were generated for this article.