Department of Human Oncology, University of Wisconsin–Madison, Madison, Wisconsin
Introduction
Patients diagnosed with localized prostate cancer are presented with multiple treatment options based on clinical and pathologic features. Conventional risk stratification with the National Comprehensive Cancer Network (NCCN) risk groups categorizes patients as low risk, intermediate risk, and high risk. National Comprehensive Cancer Network risk groups have evolved to include 6 tiers (very low risk, low risk, favorable intermediate risk, unfavorable intermediate risk, high risk, and very high risk). It is important to note that these risk groups were generated from retrospective data to provide prognostic information. These groups provide context for interpretation of clinical trial results and allow for generalized treatment recommendations as outlined by expert consensus panels. These risk groups are heterogeneous and consist of individuals at wide ranges of risk. Following these guidelines inherently results in a proportion of patients receiving excess treatment and other patients receiving undertreatment. National Comprehensive Cancer Network risk groups are outperformed by multiple newer clinical tools derived from clinical and pathologic variables as well as novel biomarkers. With the recent introduction of multiple advanced tools derived from tissue-based molecular tests (Decipher [Veracyte], Prolaris [Myriad Genetics], and Genomic Prostate Score [GPS; mdxhealth]) and digital histopathology (ArteraAI), the opportunities for precision medicine in prostate cancer have never been greater. This review aims to summarize the current data supporting these biomarkers to help clinicians select appropriate advanced tools and appropriately interpret the results of these tests for clinical decision-making.
Before any critical appraisal of the literature surrounding advanced tools for prostate cancer is undertaken, it is important to consider some fundamental aspects of biomarkers. First, the distinction between prognostic and predictive biomarkers is essential for appropriate interpretation of these tests. Prognostic biomarkers identify the likelihood of a particular end point (eg, biochemical recurrence, distant metastasis, prostate cancer–specific mortality) independent of the treatment. Predictive biomarkers identify patients who are more likely to respond favorably to a particular medical intervention or treatment compared with other similar patients. Both prognostic and predictive biomarkers are useful for informing treatment decisions for men with localized prostate cancer. For example, a prognostic biomarker could identify a patient at high risk for an adverse outcome. This patient may benefit from treatment intensification as a result of their poor prognosis. In that same patient, a predictive biomarker would help ascertain whether a specific treatment would benefit the patient.
Key Points
- Advanced tools such as Decipher, Prolaris, GPS, and ArteraAI improve risk stratification of localized prostate cancer and can help guide treatment decisions.
- Understanding the context of biomarker development, study design, end point selection, and validation is important for appropriate use and interpretation of results from advanced tools.
- Well-designed prospective biomarker studies are needed to confirm clinical utility of advanced tools across localized prostate cancer disease states.
Abbreviations
ADT androgen-deprivation therapy
AI artificial intelligence
ASCO American Society of Clinical Oncology
GPS Genomic Prostate Score
NCCN National Comprehensive Cancer Network
OS overall survival
PORTOS Prostate Cancer Radiation Therapy Outcome Score
PSA prostate-specific antigen
RT radiation therapy
RTOG Radiation Therapy Oncology Group
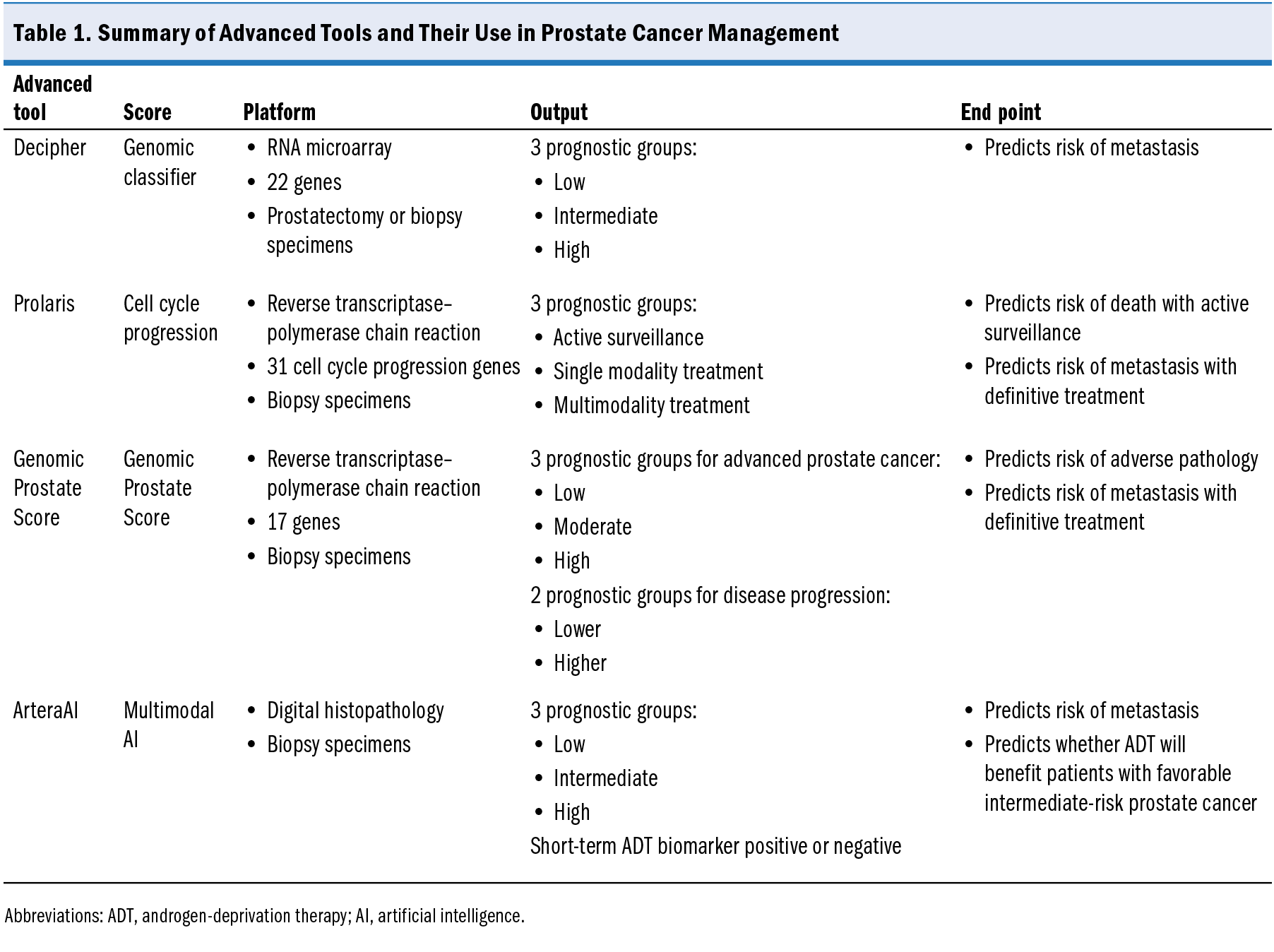
Currently commercially available tissue-based molecular tests for prostate cancer include Decipher, Prolaris, and GPS. ArteraAI is currently the only digital histopathology tool listed in NCCN guidelines. Characteristics of these tests are summarized in Table 1.
To appropriately select and use biomarkers for prostate cancer, it is critical to consider the context of the model development, including patient characteristics, study design, end point selection, analytical validity, and clinical validation. To objectively evaluate tumor marker studies, the American Society of Clinical Oncology (ASCO) developed a levels of evidence scale A to D,1 a system that was refined in 2009 to better address studies in which tissue specimens were prospectively collected in the context of a clinical trial and then retrospectively analyzed using a particular biomarker. In this new system, tumor marker studies would be classified as Simon level I in 2 different scenarios: (1) a prospective controlled trial specifically designed to address a tumor marker question or (2) archived samples from a prospective trial not designed for a tumor marker question, with 1 or more validation studies with consistent results.2 This second pathway has been used extensively for prostate cancer biomarker studies largely because of the challenges associated with prospective trials and the follow-up time necessary to generate high-level data in prostate cancer. As such, many of the studies described in this review were generated from pathology specimens collected from randomized trials that enrolled patients between 1980 and 2000. Note that Simon level I differs from category 1 evidence generated directly from a randomized control trial. To date, no advanced tool for prostate cancer has category 1 evidence, though multiple ongoing prospective trials will add stronger support for these tests once they are complete.
The ASCO issued guidance surrounding the use of these tests in 2019, stating that tissue-based genomic tests may be offered when the results are likely to affect disease management but advises against their routine use.3 Of note, the ASCO guideline lists GPS, Prolaris, and Decipher as suitable tests for localized prostate cancer management decisions but only Decipher as suitable for postprostatectomy management. No guidance has been issued by ASCO regarding the use of artificial intelligence (AI)–based digital histopathology tools.
The NCCN guidelines first included Prolaris and GPS in 2015, Decipher in 2018, and ArteraAI in 2023. As data emerged supporting the use of each test, guidelines were updated to include Simon levels I through III. The most recent guideline limits (version 1.2025) included tests for patients with Simon level I, stating that “a comprehensive list of advanced tools that do not reach the threshold of level 1 evidence is outside the scope of this guideline.” The aim of this article is to review evidence supporting the use of commercially available advanced tools for prostate cancer.
Clinical Review
Clinical scenarios discussed in this review are limited to those in which the use of an advanced tool could potentially change treatment management. Common scenarios for localized prostate cancer management will be discussed starting with the decision between treatment and surveillance in favorable-risk prostate cancer and moving on to treatment intensification decisions in higher-risk localized prostate cancer and postoperative or adjuvant treatment.
The use of these advanced tools has been increasing over time.4 A recent survey of expert consensus regarding tissue-based molecular tests revealed that more than 3 of 4 respondents believed these tools had value in risk stratification and treatment selection for patients across NCCN risk groups. Only approximately 1 in 4 experts, however, reported using genomic classifier tests clinically. Among surveyed experts, metastasis-free survival was believed to be the most relevant end point for low-risk (83%), favorable intermediate-risk (97%), and high-risk (97%) prostate cancer biomarker studies, respectively. Time to distant metastasis was believed to be a relevant end point for favorable intermediate-risk (82%) and high-risk (93%) prostate cancer biomarker studies.5 The following discussion focuses on studies with relevant end points in each clinical scenario.
The Decision Between Surveillance and Treatment
In NCCN low-risk/favorable intermediate-risk localized prostate cancer, active surveillance is a reasonable and often preferable management strategy. A prospective randomized control trial comparing active monitoring, surgery, and radiation therapy (RT) found no statistically significant difference in deaths from prostate cancer between the treatment arms, though the rate of metastases was higher in the active monitoring arm than in either treatment arm (9.4% vs 4.7% vs 5.0%).6 Tissue-based molecular and digital pathology tests may help identify patients who are otherwise low risk but are at a higher risk for metastasis and therefore less suitable for active surveillance. These tests may conversely help identify patients who are intermediate or even high risk by conventional prognostic classification but suitable for active surveillance.
Current NCCN guidelines focus on the role of advanced tools for treatment intensification in localized and postoperative treatment but do not discuss the use of these advanced tools for helping with shared decision-making surrounding active surveillance. An ideal biomarker for this clinical question would be one generated from biopsy tissue specimen (ie, treatment naive) and validated for clinically meaningful end points such as distant metastasis or prostate cancer–specific mortality. Additional end points such as likelihood of adverse pathology or time to treatment may also be of interest to patients and their clinicians and assist in the decision to manage conservatively or proceed with definitive treatment.
Among the advanced tools discussed here, Prolaris is the only test that was specifically designed in a cohort of patients with conservatively managed localized prostate cancer.7 The active surveillance threshold was validated to dichotomize patients based on 10-year risk of prostate cancer–specific mortality.8 Additional retrospective cohorts from multiple validation studies are well summarized in a meta-analysis demonstrating the prognostic utility of cell cycle progression for identifying patients at increased risk of distant metastasis and prostate cancer–specific mortality.9 A 1-unit increase in cell cycle progression was associated with a 2-fold increase in risk for death from prostate cancer.9
Other commercially available advanced tests (Decipher, GPS, ArteraAI) were not originally developed from biopsy specimens in treatment-naive patient cohorts, with data being extrapolated from retrospective studies in other clinical contexts. Based on a pooled analysis of multiple prospective studies, multimodal AI identifies patients across NCCN risk groups that have a low risk of distant metastasis and are therefore suitable for more conservative management than guideline-recommended treatment.10 In the low-risk/favorable intermediate-risk setting, patients with low multimodal AI–determined risk may be good candidates for active surveillance, whereas patients with high multimodal AI–determined risk may benefit from definitive treatment at earlier time points. The ArteraAI prognostic report now also includes the likelihood of adverse pathology in patients with low-risk/favorable intermediate-risk prostate cancer, with a validation study recently presented in abstract form.11
Multiple retrospective studies using Decipher score, primarily in patients treated with radical prostatectomy, have been published with the genomic classifier “prognostic” for a variety of oncologic end points. The use of Decipher with biopsied tissue increases the likelihood of management with active surveillance, according to a recent registry study.12 Decipher scores from biopsied tissue were correlated with adverse pathology at time of radical prostatectomy in a cohort of patients suitable for active surveillance.13 A prospective registry study associated Decipher score from biopsy with time to treatment and time to treatment failure.14 Decipher scores from prostate biopsy correlated with 10-year distant metastasis risk in a small cohort of patients treated with radical prostatectomy.15 GPS was designed and validated to predict adverse pathology at the time of prostatectomy in a population of patients suitable for active surveillance.16 Subsequent validation studies of GPS have yielded mixed results.17,18
An observational registry study compared results of tissue-based genomic tests in a population of patients largely suitable for active surveillance and found statistically significant variation in the proportion of patients who were above the test-specific threshold.19 For example, in the subgroup of patients with Gleason 3 + 3 = 6 adenocarcinoma, rates of patients above the test-specific threshold were statistically significantly lower among patients tested with Prolaris (14%) compared with patients tested with GPS (40%) or Decipher (58%). The ProtecT study reported a 9.4% rate of distant metastasis in the surveillance cohort.6 If using GPS or Decipher to make decisions regarding treatment vs active surveillance, these data suggest an excess proportion of patients may be steered toward treatment. The discordance between genomic test results reflects differences in the clinical scenarios upon which these models were generated and how test-specific thresholds were generated.
Intensification of Treatment With Definitive Radiation
When considering the addition of androgen-deprivation therapy (ADT) to RT, clinicians are faced with multiple clinical decisions, including the use or omission of ADT, the duration of ADT, the sequencing of ADT with the start of RT, and RT dose or delivery method. A recent meta-analysis combined data from 11 clinical trials investigating the addition of ADT to definitive RT and found that ADT improves overall survival (OS) and metastasis-free survival, with 1 distant metastasis prevented for every 16 patients with favorable intermediate-risk prostate cancer treated and 1 distant metastasis prevented for every 10 patients with high-risk prostate cancer treated.20 Advanced tools have the potential to better identify patients who will benefit from the addition of ADT.
Current NCCN guidelines list ArteraAI and Decipher as advanced tools with Simon level I evidence for risk stratification in patients with localized prostate cancer. Both tests have been validated using pathologic specimens banked from phase 3 randomized controlled trials. The multimodal AI score was trained from pathologic specimens from 4 phase 3 trials investigating the addition of ADT with definitive RT and validated from Radiation Therapy Oncology Group (RTOG) 9408, which randomly assigned localized patients with (mostly NCCN level favorable intermediate risk) prostate cancer to RT with or without 4 months of ADT, with biomarker-positive patients having a statistically significant reduction in risk for distant metastasis with the addition of ADT to RT; no such interaction was seen in biomarker-negative patients.21 Additional post hoc studies of multimodal AI scores from prospective trial specimens further validate the tool’s utility for stratification across NCCN risk groups.10,22,23
The evidence supporting the use of Decipher comes from numerous retrospective studies summarized in a systematic review.24 A post hoc analysis of RTOG 0126, a prospective randomized trial investigating external beam RT dose escalation without the use of ADT, provided higher-level evidence supporting the use of Decipher for prognosticating favorable intermediate-risk prostate cancer.25 Although ADT use was not an intervention studied in this trial, Decipher testing was prognostic for biochemical failure, prostate cancer–specific mortality, and distant metastasis, thereby identifying patients who would potentially benefit from treatment intensification. In the high-risk prostate cancer setting, a post hoc analysis of 3 randomized controlled trials of external beam RT (RTOG 9202, RTOG 9413, and RTOG 9902) similarly revealed that Decipher score is independently associated with distant metastasis, prostate cancer–specific mortality, and OS.26 Two active prospective randomized controlled trials, NRG GU-009 and NRG GU-010, are investigating the use of Decipher testing for treatment de-escalation and intensification in high-risk prostate cancer and unfavorable intermediate-risk prostate cancer, respectively (ClinicalTrials.gov identifiers NCT04513717 and NCT05050084). Of note, in the unfavorable intermediate-risk setting, GU-010 is investigating the omission of ADT with a Decipher score lower than 0.4 and the addition of darolutamide to standard ADT in a cohort with Decipher scores greater than 0.4. In the high-risk setting, GU-009 is similarly investigating de-escalation of ADT duration to 1 year in a cohort of patients with Decipher scores lower than 0.85 and the addition of apalutamide to the standard 2 years of ADT in a cohort with Decipher scores greater than 0.85. When completed, these studies will provide stronger evidence regarding the prognostic utility of Decipher for tailoring ADT decisions when treating localized prostate cancer with definitive RT.
The Prostate Cancer Radiation Therapy Outcome Score (PORTOS) is a 24-gene expression signature generated from the same platform as the Decipher genomic classifier; it is used to estimate radiation dose response.27 A recent validation study using samples from RTOG 0126, which studied radiation dose escalation in patients with favorable intermediate-risk prostate cancer, demonstrated that PORTOS can identify a subset of patients who may benefit from RT dose escalation.28
A cell-cycle risk score combining Prolaris cell-cycle progression score and University of California San Francisco Cancer of the Prostate Risk Assessment score has been validated as a prognostic biomarker for distant metastasis in patients receiving definitive RT.29 Patients with a cell-cycle risk score above the multimodality threshold had statistically significantly higher risk of distant metastasis than patients whose scores were below the threshold.30 A subsequent validation study illustrated that patients with cell-cycle risk scores above the multimodality threshold had statistically significantly higher rates of early metastasis when treated with single-modality therapy (RT alone or surgery) than patients receiving multimodality therapy.31 A recent study modeled the absolute risk reduction of adding ADT to RT by patient cell-cycle risk scores, ranging from 0% to 17%.32
GPS was recently studied in the context of definitive external beam RT, with a retrospective study demonstrating its prognostic utility for identifying men at risk for biochemical failure, distant metastasis, and death related to prostate cancer.33
Postoperative RT
Biochemical recurrence after definitive treatment with radical prostatectomy is a common clinical scenario. Standard salvage treatment approaches include RT to the prostate bed, with escalation options including elective nodal irradiation and the addition of concurrent or adjuvant androgen deprivation. RTOG 9601 demonstrated a benefit to the addition of 2 years of ADT with bicalutamide to salvage RT in patients with biochemical recurrence (postoperative prostate-specific antigen [PSA] level, 0.2-4.0 ng/mL), but patients with a PSA lower than 0.6 ng/mL did not have a survival benefit in a subsequent publication from this study.34,35 RTOG 0534 recently demonstrated increasing biochemical control rates with the addition of short-term ADT and pelvic nodal irradiation to prostate bed–only irradiation.36
The Decipher genomic classifier was originally generated and validated from a cohort of patients with localized prostate cancer treated with radical prostatectomy at high risk of biochemical recurrence (PSA level >20 ng/mL; Gleason score ≥8; pathologic tumor stage T3b).37,38 A validation study of the Decipher test was completed from the banked pathology specimens from the RTOG 9601 cohort with the genomic classifier independently associated with distant metastasis, prostate cancer–specific mortality, and OS.39 This study was the first one published to use pathologic specimens from a randomized controlled trial for validation of a tissue-based molecular test. The original study schema produced well-balanced groups of patients treated with and without hormone therapy. Although no statistically significant interaction was detected between Decipher and ADT treatment effect, the subset of men treated with early salvage RT (PSA level <0.7 ng/mL) with intermediate-high Decipher scores had an 11.2% absolute reduction in 12-year distant metastasis and a 4.6% absolute reduction in OS with the addition of bicalutamide. Based on this study, Decipher scores from radical prostatectomy are highly prognostic for oncologic outcomes of salvage RT, but they are not predictive of response to ADT. A recent study presented in abstract form using pathologic specimens from the RTOG 0534 study revealed that distant metastasis risk, as predicted by Decipher genomic classifier, can be mitigated by treatment escalation with the addition of elective pelvic nodal irradiation.40 As discussed earlier in this review, PORTOS has also demonstrated value for identifying patients who may benefit from RT dose escalation in the postoperative setting based on an analysis of pathologic specimens from the SAKK 09/10 study.28
Other advanced tools have retrospective data relevant to postoperative RT decisions, but they are lacking in validation from randomized controlled trials. Prolaris scores from biopsied and radical prostatectomy specimens have also been correlated with risk for distant metastasis and prostate cancer–specific mortality following radical prostatectomy in multiple retrospective studies.41-43 GPS was generated to predict adverse path at the time of radical prostatectomy, but a subsequent retrospective validation study of GPS from biopsied specimens found a strong association with prostate cancer death and metastasis.44
Conclusions
The landscape of prostate cancer management is evolving rapidly with the introduction of advanced molecular genomic and digital histopathology tools, which offer a more precise approach to risk stratification and treatment decision-making. These advanced tools provide critical prognostic and predictive insights that can refine clinical decisions in active surveillance, treatment intensification, and postoperative RT. Although consensus guidelines and expert panels help guide use of these tests, ongoing prospective trials are needed to confirm clinical utility and refine appropriate use. In the absence of prospective data, it is critical that clinicians understand the context in which these advanced tools were developed and validated to appropriately use and interpret their results.
References
1. Hayes DF, Bast RC, Desch CE, et al. Tumor marker utility grading system: a framework to evaluate clinical utility of tumor markers. J Natl Cancer Inst. 1996;88(20):1456-1466. doi:10.1093/jnci/88.20.1456
2. Simon RM, Paik S, Hayes DF. Use of archived specimens in evaluation of prognostic and predictive biomarkers. J Natl Cancer Inst. 2009;101(21):1446-1452. doi:10.1093/jnci/djp335
3. Eggener SE, Rumble RB, Armstrong AJ, et al. Molecular biomarkers in localized prostate cancer: ASCO guideline. J Clin Oncol. 2020;38(13):1474-1494. doi:10.1200/JCO.19.02768
4. Bologna E, Ditonno F, Licari LC, et al. Tissue-based genomic testing in prostate cancer: 10-year analysis of national trends on the use of Prolaris, Decipher, ProMark, and Oncotype DX. Clin Pract. 2024;14(2):508-520. doi:10.3390/clinpract14020039
5. Spohn SKB, Draulans C, Kishan AU, et al. Genomic classifiers in personalized prostate cancer radiation therapy approaches: a systematic review and future perspectives based on international consensus. Int J Radiat Oncol Biol Phys. 2023;116(3):503-520. doi:10.1016/j.ijrobp.2022.12.038
6. Hamdy FC, Donovan JL, Lane JA, et al; ProtecT Study Group. Fifteen-year outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med. 2023;388(17):1547-1558. doi:10.1056/NEJMoa2214122
7. Cuzick J, Berney DM, Fisher G, et al; Transatlantic Prostate Group. Prognostic value of a cell cycle progression signature for prostate cancer death in a conservatively managed needle biopsy cohort. Br J Cancer. 2012;106(6):1095-1099. doi:10.1038/bjc.2012.39
8. Lin DW, Crawford ED, Keane T, et al. Identification of men with low-risk biopsy-confirmed prostate cancer as candidates for active surveillance. Urol Oncol. 2018;36(6):310.e7-310.e13. doi:10.1016/j.urolonc.2018.03.011
9. Sommariva S, Tarricone R, Lazzeri M, Ricciardi W, Montorsi F. Prognostic value of the cell cycle progression score in patients with prostate cancer: a systematic review and meta-analysis. Eur Urol. 2016;69(1):107-115. doi:10.1016/j.eururo.2014.11.038
10. Tward JD, Huang HC, Esteva A, et al. Prostate cancer risk stratification in NRG oncology phase III randomized trials using multimodal deep learning with digital histopathology. JCO Precis Oncol. 2024;8:e2400145. doi:10.1200/PO.24.00145
11. Cooperberg MR, Shee K, Cowan JE, et al. Predicting long term outcomes following radical prostatectomy using a validated pathology-based multimodal artificial intelligence biomarker. J Clin Oncol. 2025;43(5_suppl):364. doi:10.1200/JCO.2025.43.5_suppl.364
12. Zaorsky NG, Proudfoot JA, Jia AY, et al. Use of the Decipher genomic classifier among men with prostate cancer in the United States. JNCI Cancer Spectr. 2023;7(5):pkad052. doi:10.1093/jncics/pkad052
13. Kim HL, Li P, Huang HC, et al. Validation of the Decipher test for predicting adverse pathology in candidates for prostate cancer active surveillance. Prostate Cancer Prostatic Dis. 2019;22(3):399-405. doi:10.1038/s41391-018-0101-6
14. Vince RA Jr, Jiang R, Qi J, et al. Impact of Decipher Biopsy testing on clinical outcomes in localized prostate cancer in a prospective statewide collaborative. Prostate Cancer Prostatic Dis. 2022;25(4):677-683. doi:10.1038/s41391-021-00428-y
15. Klein EA, Haddad Z, Yousefi K, et al. Decipher genomic classifier measured on prostate biopsy predicts metastasis risk. Urology. 2016;90:148-152. doi:10.1016/j.urology.2016.01.012
16. Klein EA, Cooperberg MR, Magi-Galluzzi C, et al. A 17-gene assay to predict prostate cancer aggressiveness in the context of Gleason grade heterogeneity, tumor multifocality, and biopsy undersampling. Eur Urol. 2014;66(3):550-560. doi:10.1016/j.eururo.2014.05.004
17. Nyame YA, Grimberg DC, Greene DJ, et al. Genomic scores are independent of disease volume in men with favorable risk prostate cancer: implications for choosing men for active surveillance. J Urol. 2018;199(2):438-444. doi:10.1016/j.juro.2017.09.077
18. Lin DW, Zheng Y, McKenney JK, et al. 17-gene genomic prostate score test results in the Canary Prostate Active Surveillance Study (PASS) cohort. J Clin Oncol. 2020;38(14):1549-1557. doi:10.1200/JCO.19.02267
19. Hu JC, Tosoian JJ, Qi J, et al. Clinical utility of gene expression classifiers in men with newly diagnosed prostate cancer. JCO Precis Oncol. 2018;2:PO.18.00163. doi:10.1200/po.18.00163
20. Kishan AU, Sun Y, Hartman H, et al; MARCAP Consortium group. Androgen deprivation therapy use and duration with definitive radiotherapy for localised prostate cancer: an individual patient data meta-analysis. Lancet Oncol. 2022;23(2):304-316. doi:10.1016/S1470-2045(21)00705-1
21. Spratt DE, Liu VYT, Yamashita R, et al. Patient-level data meta-analysis of a multi-modal artificial intelligence (MMAI) prognostic biomarker in high-risk prostate cancer: results from six NRG/RTOG phase III randomized trials. J Clin Oncol. 2023;41(6_suppl):299. doi:10.1200/JCO.2023.41.6_suppl.299
22. Esteva A, Feng J, van der Wal D, et al. Prostate cancer therapy personalization via multi-modal deep learning on randomized phase III clinical trials. NPJ Digit Med. 2022;5(1):71. doi:10.1038/s41746-022-00613-w
23. Ross AE, Zhang J, Huang HC, et al. External validation of a digital pathology-based multimodal artificial intelligence architecture in the NRG/RTOG 9902 phase 3 trial. Eur Urol Oncol. 2024;7(5):1024-1033. doi:10.1016/j.euo.2024.01.004
24. Jairath NK, Dal Pra A, Vince R Jr, et al. A systematic review of the evidence for the Decipher genomic classifier in prostate cancer. Eur Urol. 2021;79(3):374-383. doi:10.1016/j.eururo.2020.11.021
25. Spratt DE, Liu VYT, Michalski J, et al. Genomic classifier performance in intermediate-risk prostate cancer: results from NRG Oncology/RTOG 0126 randomized phase 3 trial. Int J Radiat Oncol Biol Phys. 2023;117(2):370-377. doi:10.1016/j.ijrobp.2023.04.010
26. Nguyen PL, Huang HCR, Spratt DE, et al. Analysis of a biopsy-based genomic classifier in high-risk prostate cancer: meta-analysis of the NRG Oncology/Radiation Therapy Oncology Group 9202, 9413, and 9902 phase 3 randomized trials. Int J Radiat Oncol Biol Phys. 2023;116(3):521-529. doi:10.1016/j.ijrobp.2022.12.035
27. Zhao SG, Chang SL, Spratt DE, et al. Development and validation of a 24-gene predictor of response to postoperative radiotherapy in prostate cancer: a matched, retrospective analysis. Lancet Oncol. 2016;17(11):1612-1620. doi:10.1016/S1470-2045(16)30491-0
28. Dal Pra A, Ghadjar P, Ryu HM, et al. Predicting dose response to prostate cancer radiotherapy: validation of a radiation signature in the randomized phase III NRG/RTOG 0126 and SAKK 09/10 trials. Ann Oncol. 2025;S0923-7534(25)00025-0. doi:10.1016/j.annonc.2025.01.017
29. Tward J, Lenz L, Flake DD II, et al. The clinical cell-cycle risk (CCR) score is associated with metastasis after radiation therapy and provides guidance on when to forgo combined androgen deprivation therapy with dose-escalated radiation. Int J Radiat Oncol Biol Phys. 2022;113(1):66-76. doi:10.1016/j.ijrobp.2021.09.034
30. Tward JD, Schlomm T, Bardot S, et al. Personalizing localized prostate cancer: validation of a combined clinical cell-cycle risk (CCR) score threshold for prognosticating benefit from multimodality therapy. Clin Genitourin Cancer. 2021;19(4):296-304.e3. doi:10.1016/j.clgc.2021.01.003
31. Hutten RJ, Odei B, Johnson SB, Tward JD. Validation of the combined clinical cell-cycle risk score to prognosticate early prostate cancer metastasis from biopsy specimens and comparison with other routinely used risk classifiers. JCO Precis Oncol. 2024;8:e2300364. doi:10.1200/PO.23.00364
32. Tward JD, Lenz L, Gutin A, et al. Using the cell-cycle risk score to predict the benefit of androgen-deprivation therapy added to radiation therapy in patients with newly diagnosed prostate cancer. JCO Precis Oncol. 2024;8:e2300722. doi:10.1200/PO.23.00722
33. Janes JL, Boyer MJ, Bennett JP, et al. The 17-gene genomic prostate score test is prognostic for outcomes after primary external beam radiation therapy in men with clinically localized prostate cancer. Int J Radiat Oncol Biol Phys. 2023;115(1):120-131. doi:10.1016/j.ijrobp.2022.06.101
34. Shipley WU, Seiferheld W, Lukka HR, et al; NRG Oncology RTOG. Radiation with or without antiandrogen therapy in recurrent prostate cancer. N Engl J Med. 2017;376(5):417-428. doi:10.1056/NEJMoa1607529
35. Dess RT, Sun Y, Jackson WC, et al. Association of presalvage radiotherapy PSA levels after prostatectomy with outcomes of long-term antiandrogen therapy in men with prostate cancer. JAMA Oncol. 2020;6(5):735-743. doi:10.1001/jamaoncol.2020.0109
36. Pollack A, Karrison TG, Balogh AG, et al. The addition of androgen deprivation therapy and pelvic lymph node treatment to prostate bed salvage radiotherapy (NRG Oncology/RTOG 0534 SPPORT): an international, multicentre, randomised phase 3 trial. Lancet. 2022;399(10338):1886-1901. doi:10.1016/S0140-6736(21)01790-6
37. Karnes RJ, Bergstralh EJ, Davicioni E, et al. Validation of a genomic classifier that predicts metastasis following radical prostatectomy in an at risk patient population. J Urol. 2013;190(6):2047-2053. doi:10.1016/j.juro.2013.06.017
38. Cooperberg MR, Davicioni E, Crisan A, Jenkins RB, Ghadessi M, Karnes RJ. Combined value of validated clinical and genomic risk stratification tools for predicting prostate cancer mortality in a high-risk prostatectomy cohort. Eur Urol. 2015;67(2):326-333. doi:10.1016/j.eururo.2014.05.039
39. Feng FY, Huang HC, Spratt DE, et al. Validation of a 22-gene genomic classifier in patients with recurrent prostate cancer: an ancillary study of the NRG/RTOG 9601 randomized clinical trial. JAMA Oncol. 2021;7(4):544-552. doi:10.1001/jamaoncol.2020.7671
40. Pollack A, Johnson M, Proudfoot J, et al. Decipher score as a predictor of response to treatment intensification in the NRG Oncology-RTOG 0534 (SPPORT) phase III randomized post-prostatectomy salvage radiotherapy trial. J Clin Oncol. 2025;43(5_suppl):399. doi:10.1200/JCO.2025.43.5_suppl.399
41. Bishoff JT, Freedland SJ, Gerber L, et al. Prognostic utility of the cell cycle progression score generated from biopsy in men treated with prostatectomy. J Urol. 2014;192(2):409-414. doi:10.1016/j.juro.2014.02.003
42. Koch MO, Cho JS, Kaimakliotis HZ, et al. Use of the cell cycle progression (CCP) score for predicting systemic disease and response to radiation of biochemical recurrence. Cancer Biomark. 2016;17(1):83-88. doi:10.3233/CBM-160620
43. Swanson GP, Lenz L, Stone S, Cohen T. Cell-cycle risk score more accurately determines the risk for metastases and death in prostatectomy patients compared with clinical features alone. Prostate. 2021;81(4):261-267. doi:10.1002/pros.24103
44. Van Den Eeden SK, Lu R, Zhang N, et al. A biopsy-based 17-gene genomic prostate score as a predictor of metastases and prostate cancer death in surgically treated men with clinically localized disease. Eur Urol. 2018;73(1):129-138. doi:10.1016/j.eururo.2017.09.013
Article Information
Published: March 31, 2025.
Conflict of Interest Disclosures: Dr Hutten received honoraria from Myriad Genetics.
Funding/Support: None.
Author Contributions: Dr Hutten is responsible for the conceptualization, investigation, data curation, and writing (original draft and review/editing).
Data Availability Statement: No new data were generated for this article.