Introduction
Regardless of disease stage, transurethral resection of a bladder tumor (TURBT) is the cornerstone of diagnosis, treatment, and risk stratification. Despite the importance of performing a technically sound and complete TURBT, the quality of TURBT varies widely among practicing urologists, potentially affecting patient outcomes. Bladder cancer is the sixth most common cancer diagnosed in the United States and was responsible for 16 840 deaths in 2024.1 Among newly diagnosed bladder cancer cases, non–muscle-invasive bladder cancer (NMIBC) accounts for 75% to 85% of cases.2 Superficial bladder cancer has a long natural history, necessitating extended risk-adapted surveillance approaches to assess disease recurrence and progression. This need results in substantial health care–related costs, patient morbidity, and inconvenience. Even with adequate treatment, studies have shown that up to 50% of high-grade NMIBC may progress to MIBC.3 Patients with MIBC are at high risk of systemic progression and death from their disease, and urgent multimodality therapy, including a maximal TURBT, is the current standard of care.
Key Points
- Standardizing TURBT is essential to providing consistent and reproducible results and optimizing surgical outcomes.
- Repeat TURBT continues to be recommended in the management of NMIBC, particularly in patients with T1 cancer.
- Novel technologies, including blue-light cystoscopy and MRI, are excellent tools for providing maximal benefit and patient-specific care.
- Maximal TURBT in the setting of trimodal therapy is a reasonable alternative to radical cystectomy for MIBC in terms of survival and oncologic outcomes, especially in older or frail patients.
Abbreviations
AI artificial intelligence
MRI magnetic resonance imaging
NMIBC non–muscle-invasive bladder cancer
TURBT transurethral resection of a bladder tumor
Historical attempts to define a “complete TURBT” and develop quality metrics that standardize the surgical approach have been inadequate. In addition, documentation regarding resection completeness and adherence to previous guidelines is often inconsistent, making the postoperative assessment of resection quality difficult to assess accurately.4 In this review, we describe contemporary approaches to standardize TURBT quality, discuss the role of novel technologies in improving surgical outcomes, analyze the importance of maximal TURBT as the primary treatment for NMIBC, and investigate the role of maximal TURBT in patients receiving trimodal therapy for MIBC.
Defining a Good or Adequate TURBT
Despite the wide variability in TURBT quality, a broad consensus exists that all visible tumors should be resected at the time of TURBT. The European Association of Urology provides detailed guidelines for TURBT techniques, recommending en bloc resection for small papillary tumors, whereas larger tumors should be resected in fractions to include adjacent bladder mucosa and deeper detrusor muscle layers and to avoid the risk of perforation.5
Residual tumor burden is closely tied to the initial tumor risk stratification. A 2022 study found that the likelihood of residual tumor was statistically significantly higher in patients with unfavorable risk profiles: 50% in the high-risk group, 18.6% in the intermediate-risk group, and 7.8% in the low-risk group.6 Furthermore, the primary tumor base and resection margins were identified as the most common sites for residual tumors, with rates of 34.4% and 50%, respectively.6 These findings raise questions about the efficacy of the initial TURBT and the necessity for re-resection, particularly in cases where maximal TURBT may not have been feasible because of tumor size or complexity. These data also underscore the importance of maintaining a high index of suspicion for residual disease in high-risk cases and the role of routine re-TURBT in this patient population.
Although neither the National Comprehensive Cancer Network nor the American Urological Association provide explicit quality standards for bladder tumor resection, re-TURBT is recommended for patients with pT1 disease. Re-TURBT increases staging accuracy, particularly in cases of persistent tumor or when detrusor muscle was absent in the initial resection. The European Association of Urology emphasizes that repeat TURBT can increase recurrence-free survival, progression-free survival, and overall survival in BCG-treated T1 high-grade/grade 3 cancer without muscle on the initial specimen.7 The absence of residual tumor on repeat resection was also linked to improved survival outcomes in patients ultimately treated with radical cystectomy.8 This repeat procedure is critical for determining the full extent of disease.
Although complete resection is desired in all TURBTs, different factors may influence the surgical complexity of endoscopic resection. Roumiguié et al9 developed a predictive model of clinicopathologic factors that, if present, may increase procedure difficulty. These factors included the patient’s past medical history (including history of radiation or previous TURBT), tumor number, location, size, and access to the bladder as independent predictors of an increasingly complex TURBT. An individual risk-adapted approach based on the features described by Roumiguié et al may help select patients who most benefit from relook cystoscopy and subsequent re-TURBT.
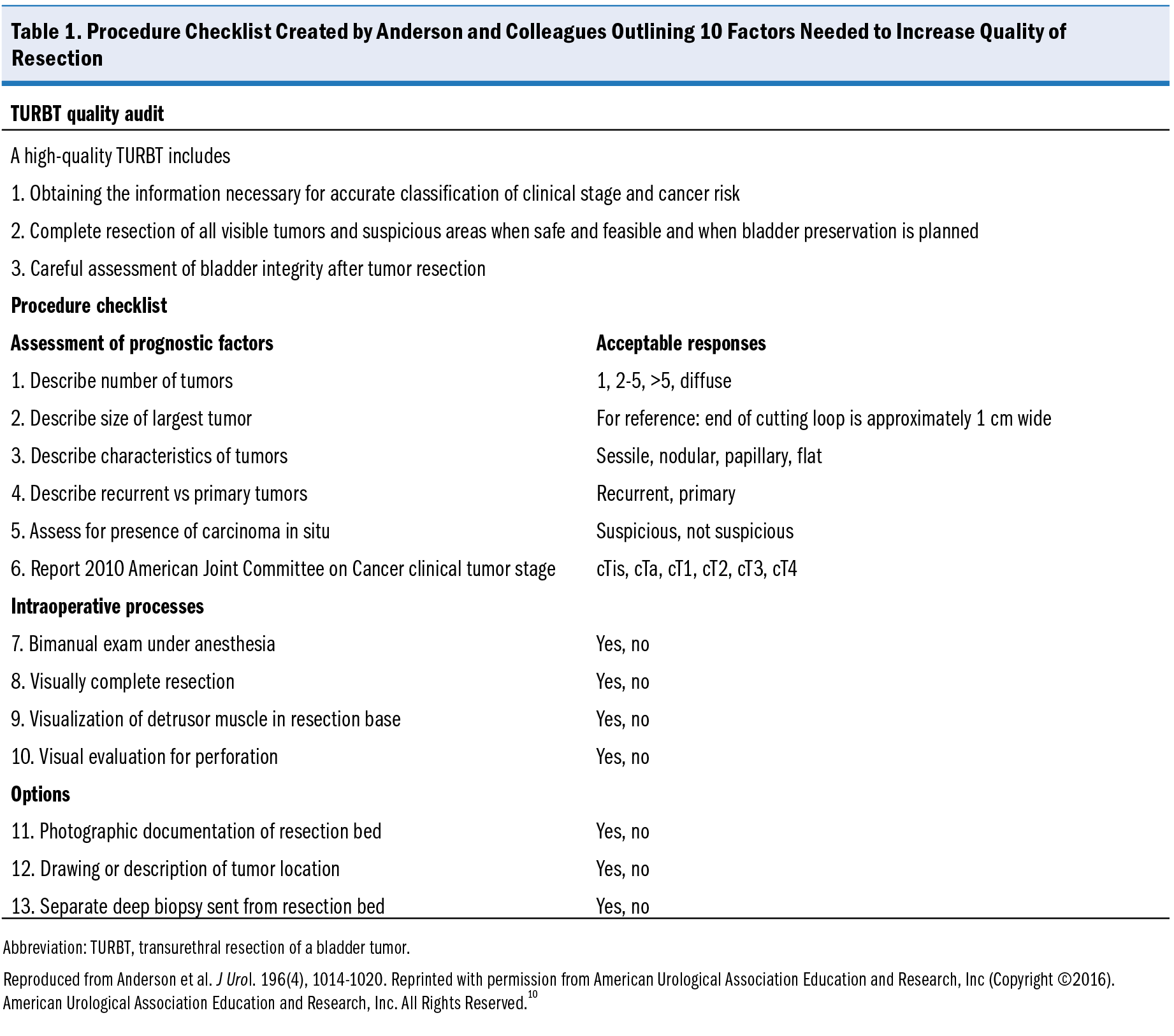
Standardizing the approach to TURBTs, especially in complex cases, is necessary to achieve reproducible results and optimize surgical outcomes. Establishing a checklist is one approach to standardization. Anderson et al10 outlined 10 critical items that should be performed with every TURBT. Items on this checklist included documenting the number and size of tumors, presence of carcinoma in situ, visualization of detrusor muscle, and complete resection of all visible tumors. Routine use of a checklist was found to substantially increase the quality of the resection and provide objective, patient-specific data to subsequent urologists to assess procedure quality (Table 1).10
Another approach to improving the quality and reproducibility of TURBT has been the use of urologist-focused quality improvement programs. Mariappan et al11 reported on their experience using urologist-focused quality improvement programs to improve TURBT quality. Their focus was aimed at educating urologists on their TURBT primary end points. They observed that quality improvement education about procedure-related quality metrics was associated with a clinically significant increase in restaging TURBT, improved tumor staging, and decreased recurrence rates.
Novel approaches to standardization and reproducibility of TURBT, including the use of virtual reality surgical simulators, have been linked to improved surgical outcomes. Bube et al12 issued a virtual reality simulator to urologists and assessed TURBT performance scores both before and after installation. After virtual reality training, novices substantially improved their TURBT performance scores, which included a more complete resection of tissue, improved hemostasis, and more rapid progression through the case. A transurethral resection model offered by Berridge et al13 found that the use of a virtual reality simulator increased confidence in their trainees. Another study had a similar finding: 87% of participants felt that their confidence in performing TURBTs increased, and 92% wanted virtual reality models in their training.14 Virtual reality surgical simulators can be applied to all levels of urologic training. For instance, Neumann et al15 analyzed the impact of virtual reality TURBT training models for medical students. The authors found substantial improvements in procedure length, resectoscope movement, and accidental bladder injury after virtual reality training in this medical student cohort. A similar virtual reality training module was analyzed in a systematic review that reported improvements in blood loss, tumor resection, and procedural time after training.16 These data collectively suggest that virtual reality surgical simulators may be a good training tool for urologists to acquire and maintain their endoscopic surgical skills.
Repeat TURBT
Repeat TURBT is universally recommended in clinical guidelines for patients with T1 NMIBC. This re-resection serves a therapeutic role in improving cancer control and provides critical staging information for determining appropriate subsequent treatment. Although repeat TURBT can be costly and carries an increased risk of complications, such as bleeding and perforation, it remains an essential step in the diagnosis and management of bladder cancer. Objectively assessing the quality of an initial resection and the associated clinicopathologic factors will identify which patients are most likely to benefit from repeat TURBT. Factors such as incomplete resection, a lack of detrusor muscle in the specimen, tumor stage, and grade determine the need to consider repeat TURBT. Improvements in endoscopic visibility and enhancement imaging techniques have improved the diagnosis of bladder cancer with adequate staging, resulting in better outcomes with lower rates of residual tumor burden.17
Studies have shown repeat resection 2 to 6 weeks after initial TURBT for high grade Ta or T1 NMIBC reveals residual tumor in up to 70% of cases and upstaging in up to 20%.18 Recurrence and upstaging rates can be secondary to inadequate specimen collection but also result from the presence of multifocal tumors and tumors presenting at a more advanced stage. Yuk et al19 found that recurrent gross hematuria, tumor grade, quantity, and size were all strongly associated with residual tumor after initial resection. A prospective randomized trial found that repeat TURBT in patients with T1 tumors increased recurrence-free and progression-free survival.20 Sharma et al21 further supported this claim, revealing that re-TURBT is essential in high-grade pTa and pT1 disease for accurate staging and management. In addition, only 2% of patients who underwent repeat TURBT died from urothelial carcinoma compared with 11% in the single-TURBT group.20,22
Although repeat TURBT is widely recognized as a critical component of adequate bladder cancer staging, recent advancements in visually enhanced TURBT and en bloc resection techniques have emerged. Over the past few decades, the rate of re-TURBT has declined, with fewer patients undergoing the procedure in the 2010s than in earlier decades. Despite this shift, patients who underwent re-TURBT demonstrated higher recurrence-free survival, with a hazard ratio of 0.78 (95% CI, 0.62-0.97), although no notable benefit was observed in progression-free survival or cancer-specific survival.23 Nevertheless, re-TURBT remains essential for accurate staging and tumor control in patients with T1 NMIBC. A shared decision-making approach should be adopted, taking into account the potential risks, including a higher complication rate associated with the repeat procedure.
Strategies to Augment TURBT Effectiveness and Risk Stratification
Emerging technologies, such as urinary cell-free tumor DNA, offer promising advancements in detecting minimal residual disease before re-TURBT in patients with high-risk NMIBC. Preliminary data from prospective studies demonstrate that tumor fraction, as assessed by urinary cell-free tumor DNA and estimated copy number burden, can effectively identify residual disease.24 This innovation challenges the routine need for re-TURBT in certain cases, such as patients with pT1 bladder cancer. One study reviewed the utility of measuring varying urine messenger RNA sequences in patients diagnosed with NMIBC vs healthy individuals. The authors found statistically significantly higher expression levels of particular messenger RNA sequences, including CA9 and CDK1, in urine samples from patients with NMIBC.25
Although no single urinary biomarker has proven optimal for improving NMIBC prognosis, a synergistic approach combining these biomarkers with maximal TURBT shows promise in predicting recurrence. Currently, US Food and Drug Administration–approved tests for diagnosis and surveillance include urinary cytology, BTA stat (Polymedco), BTA TRAK (Polymedco), nuclear matrix protein, and UroVysion fluorescence in situ hybridization (Abbott Molecular). ImmunoCyt (DiagnoCure) has been specifically approved for surveillance purposes.26 Elsawy et al27 used a urinary messenger RNA biomarker, Xpert Test (Cepheid), after initial resection of T1 bladder cancer. The authors found that a positive Xpert Test after complete resection of T1 disease was associated with positive repeat biopsy, along with early tumor recurrence.
In addition to biomarkers, advanced visual technologies continue to increase diagnostic accuracy for bladder cancer, further supporting the goal of tailored and effective patient care. Narrow-band imaging (an optic enhancement technique that uses various bandwidths of light to further delineate tissue) and blue-light cystoscopy can be used to reduce recurrence rates of NMIBC after TURBT.
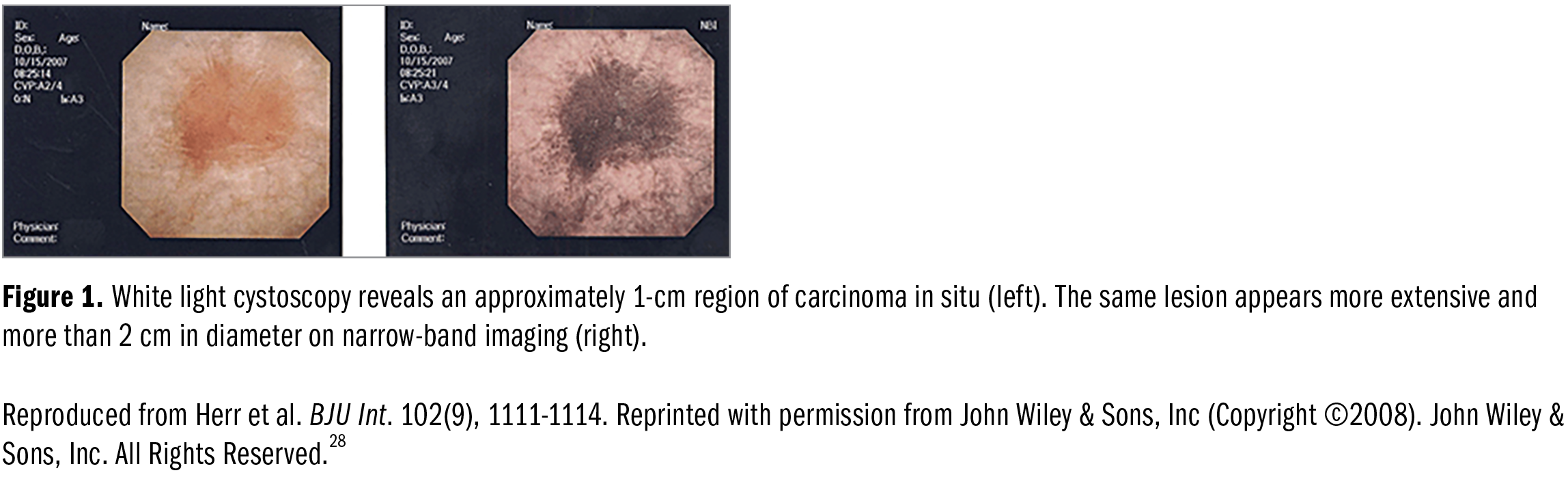
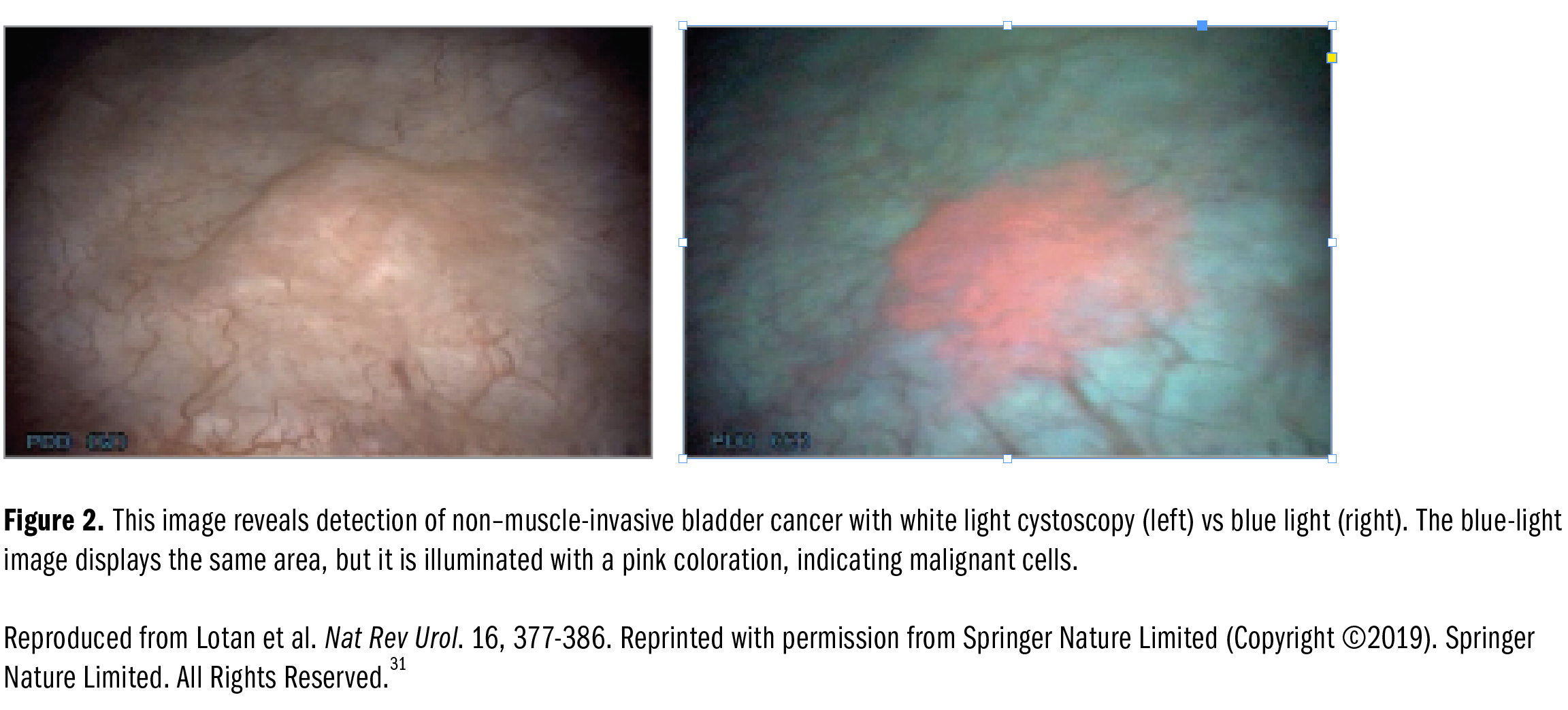
Herr et al28 found that 24% of patients with NMIBC had recurrent cancer after TURBT. Of these cancers, 87% were diagnosed on white-light cystoscopy and narrow-band imaging, and 100% were detected on narrow-band imaging cystoscopy. Narrow-band imaging cystoscopy was more effective in detecting papillary tumors and carcinoma in situ (Figure 1). Another study showed that narrow-band imaging was effective in identifying 36 (17.1%) tumors in patients, whereas white light found only 4 (1.9%) tumors.29 Blue-light cystoscopy involves intravesical administration of hexaminolevulinate, a molecule that is incorporated into heme biosynthesis and results in an accumulation in malignant cells.30 This bioluminescence helps delineate bladder cancer cells for a more complete resection (Figure 2).31 Denzinger et al32 found that in 301 patients with superficial bladder cancer, the residual tumor rate was 25.2% in the white-light cystoscopy group and 4.5% in the fluorescence cystoscopy group. Recurrence-free survival was higher in the fluorescence cystoscopy group up to 8 years after TURBT follow-up.32 Carcinoma in situ was also more readily diagnosed with blue-light cystoscopy than with traditional white light.33 Tadrist et al34 report that residual tumor was detected in 64 of 109 (58.7%) patients at second TURBT on photodynamic diagnosis after a primary blue-light resection, supporting the need for restage TURBT, even with photodynamic diagnosis.
Magnetic resonance imaging (MRI) has also been described as an effective supplement in management of bladder cancer with TURBT. A systematic review analyzed the implications of an integrating multiparametric MRI and the Vesical Imaging Reporting and Data System in bladder cancer staging—specifically, on restaging TURBT. The authors found that multiparametric MRI and the Vesical Imaging Reporting and Data System may be effective tools in identifying patients who may benefit from restaging TURBT.35 Another study performed post-TURBT bladder MRIs and found a 78.9% accuracy rate of MRI in patients without residual tumor.36 Although they have not yet been adopted in the European Association of Urology guidelines, new trials, including the 2024 CUT-less trial, aim to include information from the Vesical Imaging Reporting and Data System and photodynamic diagnosis in restaging TURBT indications.
Artificial intelligence (AI) has been investigated as effective adjunct to TURBT, and AI is a promising new frontier in therapeutic interventions for bladder cancer. Examples include 3-dimensional reconstruction models that include texture modeling and simulation.37 In addition, augmented cystoscopy through AI could assist in determining tumor location, size, and other characteristics. Compared with expert urologists, AI demonstrated higher classification sensitivity and specificity for tumor grade and malignancy.38 Artificial intelligence can also be applied to predicting recurrence in NMIBC. Abbas et al39 found that AI can predict NMIBC recurrence in up to 97.5% of cases, although the current average is approximately 75%. Another study incorporated multiparametric MRIs and clinical factors into a deep learning model to predict 5-year recurrence in NMIBC. The authors found superior performance in this deep learning model for analyzing recurrence, although additional studies must be performed.40
Frailty and Older Patients in Maximal TURBT
As the global population ages, the issue of frailty in urological procedures has become a critical concern, particularly in the context of shared decision-making for invasive interventions. Frailty is increasingly relevant in cancer care, given the aging population. For instance, the average age at diagnosis for bladder cancer is 73 years, notably older than the mean age for all cancer diagnoses.41 Although frailty is not synonymous with functional impairment, it is a factor influencing postoperative outcomes in geriatric urologic patients. Studies have shown that frailty and advanced age are associated with heightened risks of postoperative complications, including delirium, intensive care unit admissions, prolonged hospital stays, and mortality.42
In the management of MIBC, the frailty index plays a pivotal role in treatment planning. Patients with higher frailty scores may be less suited for radical cystectomy and instead may benefit from trimodal therapy, which includes maximal TURBT. For example, older age (≥70 years) has been linked to statistically significantly higher 1-year mortality in patients who underwent radical cystectomy.43 Although survival outcomes between carefully selected patients undergoing radical cystectomy and trimodal therapy are comparable, there is no established frailty-based selection criterion for either treatment approach. Notably, frailty prevalence is statistically significantly higher in patients opting for trimodal therapy (56%) compared with patients undergoing radical cystectomy (6.2%).44 In fact, some studies have demonstrated improved long-term quality of life after trimodal therapy vs radical cystectomy. In the 4 Radiation Therapy Oncology Group trials, no patients needed radical cystectomy for treatment-related toxicity or experienced treatment-related mortality.45
In this aging population, treatment strategies must prioritize minimizing postoperative complications while achieving an optimal balance between cancer control and quality of life. For patients with a higher frailty index, maximal TURBT combined with trimodal therapy is often better tolerated than the more invasive radical cystectomy. Five-year survival rates in trimodal therapy have been found to be as high as 65%, similar to rates in radical cystectomy.46 Disease-specific survival was also nearly identical, regardless of age, further emphasizing trimodal therapy as an effective alternative to radical cystectomy in MIBC.47 Effective shared decision-making, incorporating frailty assessments, is essential to achieving the best possible outcomes for these patients.
Conclusion
Maximal TURBT in NMIBC remains the best opportunity to treat and risk stratify patients with bladder cancer. Standardized approaches and guideline-based restaging TURBT aim to maximize cure rates while decreasing the chances of progression and disease recurrence. Properly performed TURBT in the setting of trimodal therapy can be applied to specific patient populations without reducing treatment efficacy. The implementation of novel technologies, including blue light, narrow-band imaging, and MRI as adjuncts to TURBT, increases the completeness of resection and may provide critical staging information for patients. Further studies are needed to assess the role of virtual reality and AI in universal protocols for NMIBC management and quality improvement initiatives.
References
1. Cancer stat facts: common cancer sites. National Cancer Institute: Surveillance, Epidemiology, and End Results Program. January 31, 2024. Accessed January 30, 2025. https://seer.cancer.gov/statfacts/html/common.html
2. Dobruch J, Oszczudłowski M. Bladder cancer: current challenges and future directions. Medicina. 2021;57(8):749. doi:10.3390/medicina57080749
3. Carrasco R, Izquierdo L, van der Heijden AG, et al. Differential gene expression profile between progressive and de novo muscle invasive bladder cancer and its prognostic implication. Sci Rep. 2021;11(1):6132. doi:10.1038/s41598-021-85137-1
4. Khattak MA, Bangash M, Aziz W, et al. Evaluating the quality of primary transurethral resection of bladder tumor: a nine-year review at a tertiary healthcare center. Cureus. 2024;16(8):e68143. doi:10.7759/cureus.68143
5. Woldu SL, Bagrodia A, Lotan Y. Guideline of guidelines: non‐muscle‐invasive bladder cancer. BJU Int. 2017;119(3):371-380. doi:10.1111/bju.13760
6. Fan J, Zhang X, Fan J, Li L, He D, Wu K. Risk stratification for the rate and location of residual bladder tumor for the decision of re-transurethral resection of bladder tumor. Front Oncol. 2022;12:788568. doi:10.3389/fonc.2022.788568
7. Gontero P, Sylvester R, Pisano F, et al. The impact of re-transurethral resection on clinical outcomes in a large multicentre cohort of patients with T1 high-grade/grade 3 bladder cancer treated with bacille Calmette-Guérin. BJU Int. 2016;118(1):44-52. doi:10.1111/bju.13354
8. Bree KK, Kokorovic A, Westerman ME, et al. Repeat transurethral resection of muscle-invasive bladder cancer prior to radical cystectomy is prognostic but not therapeutic. J Urol. 2023;209(1):140-149. doi:10.1097/JU.0000000000003015
9. Roumiguié M, Xylinas E, Brisuda A, et al. Consensus definition and prediction of complexity in transurethral resection or bladder endoscopic dissection of bladder tumours. Cancers. 2020;12(10):3063. doi:10.3390/cancers12103063
10. Anderson C, Weber R, Patel D, et al. A 10-item checklist improves reporting of critical procedural elements during transurethral resection of bladder tumor. J Urol. 2016;196(4):1014-1020. doi:10.1016/j.juro.2016.03.151
11. Mariappan P, Johnston A, Padovani L, et al. Enhanced quality and effectiveness of transurethral resection of bladder tumour in non-muscle-invasive bladder cancer: a multicentre real-world experience from Scotland’s Quality Performance Indicators Programme. Eur Urol. 2020;78(4):520-530. doi:10.1016/j.eururo.2020.06.051
12. Bube SH, Kingo PS, Madsen MG, et al. National implementation of simulator training improves transurethral resection of bladder tumours in patients. Eur Urol Open Sci. 2022;39:29-35. doi:10.1016/j.euros.2022.03.003
13. Berridge C, Kailavasan M, Athanasiadis G, et al. Endoscopic surgical simulation using low-fidelity and virtual reality transurethral resection simulators in urology simulation boot camp course: trainees feedback assessment study. World J Urol. 2021;39(8):3103-3107. doi:10.1007/s00345-020-03559-4
14. Moore J, Whalen S, Rowe N, Lee JY, Ordon M, Lantz Powers AG. A high-fidelity, virtual-reality, transurethral resection of bladder tumor simulator: validation as a tool for training. Can Urol Assoc J. 2022;16(4):E205-E211. doi:10.5489/cuaj.7285
15. Neumann E, Mayer J, Russo GI, et al. Transurethral resection of bladder tumors: next-generation virtual reality training for surgeons. Eur Urol Focus. 2019;5(5):906-911. doi:10.1016/j.euf.2018.04.011
16. Kallidonis P, Peteinaris A, Ortner G, et al. Transurethral resection of bladder tumor: a systematic review of simulator-based training courses and curricula. Asian J Urol. 2024;11(1):1-9. doi:10.1016/j.ajur.2022.08.005
17. Yanagisawa T, Sato S, Hayashida Y, et al. Do we need repeat transurethral resection after en bloc resection for pathological T1 bladder cancer?. BJU Int. 2023;131(2):190-197. doi:10.1111/bju.15760
18. Alsyouf M, Ladi-Seyedian SS, Konety B, et al. Is a restaging TURBT necessary in high-risk NMIBC if the initial TURBT was performed with blue light? Urol Oncol. 2023;41(2):109.e9-109.e14. doi:10.1016/j.urolonc.2022.10.026
19. Yuk HD, Kim JK, Jeong CW, Kwak C, Kim HH, Ku JH. Differences in pathologic results of repeat transurethral resection of bladder tumor (TURBT) according to institution performing the initial TURBT: comparative analyses between referred and nonreferred group. Biomed Res Int. 2018;2018:1-7. doi:10.1155/2018/9432606
20. Divrik RT, Sahin AF, Yildirim U, Altok M, Zorlu F. Impact of routine second transurethral resection on the long-term outcome of patients with newly diagnosed pT1 urothelial carcinoma with respect to recurrence, progression rate, and disease-specific survival: a prospective randomised clinical trial. Eur Urol. 2010;58(2):185-190. doi:10.1016/j.eururo.2010.03.007
21. Sharma A, Raghavendra RT, Biswal D, Yadav P, Goel S, Sharma S. Rationale of restaging transurethral resection of bladder tumor in patients with nonmuscle invasive bladder cancer in the current era. Urol Ann. 2024;16(4):288-291. doi:10.4103/ua.ua_50_24
22. Herr HW. Role of repeat resection in non-muscle-invasive bladder cancer. J Natl Compr Canc Netw. 2015;13(8):1041-1046. doi:10.6004/jnccn.2015.0123
23. Yanagisawa T, Kawada T, Von Deimling M, et al. Repeat transurethral resection for non-muscle invasive bladder cancer: an updated systematic review and meta-analysis in the contemporary era. Eur Urol Focus. 2024;10(1):41-56. doi:10.1016/s0302-2838(24)01305-8
24. Linscott JA, Miyagi H, Murthy PB, et al. From detection to cure—emerging roles for urinary tumor DNA (utDNA) in bladder cancer. Curr Oncol Rep. 2024;26(8):945-958. doi:10.1007/s11912-024-01555-0
25. Abazari O, Shahidi M, Dayati P, et al. Study of urine-based mRNA biomarkers for early detection of nonmuscle invasive bladder cancer (NMIBC). Urol Oncol. 2025;11:S1078-1439(24)01050-0. doi:10.1016/j.urolonc.2024.12.273
26. Darwiche F, Parekh DJ, Gonzalgo ML. Biomarkers for non-muscle invasive bladder cancer: current tests and future promise. Indian J Urol. 2015;31(4):273-282. doi:10.4103/0970-1591.166448
27. Elsawy AA, Awadalla A, Abdullateef M, Ahmed AE, Abol-Enein H. Can repeat biopsy be skipped after initial complete resection of T1 bladder cancer? The role of a novel urinary mRNA biomarker. Urol Oncol. 2021;39(7):437.e11-437.e19. doi:10.1016/j.urolonc.2021.02.009
28. Herr HW, Donat SM. A comparison of white-light cystoscopy and narrow-band imaging cystoscopy to detect bladder tumour recurrences. BJU Int. 2008;102:1111-1114. doi:10.1111/j.1464-410X.2008.07846.x
29. Shen YJ, Zhu YP, Ye DW, et al. Narrow-band imaging flexible cystoscopy in the detection of primary non-muscle invasive bladder cancer: a “second look” matters? Int Urol Nephrol. 2012;44(2):451-457. doi:10.1007/s11255-011-0036-5
30. Cahill EM, Chua K, Doppalapudi SK, Ghodoussipour S. The use of blue-light cystoscopy in the detection and surveillance of nonmuscle invasive bladder cancer. Curr Urol. 2022;16(3):121-126. doi:10.1097/CU9.0000000000000142
31. Lotan Y, Bivalacqua TJ, Downs T, et al. Blue light flexible cystoscopy with hexaminolevulinate in non-muscle-invasive bladder cancer: review of the clinical evidence and consensus statement on optimal use in the USA—update 2018. Nat Rev Urol. 2019;16(6):377-386. doi:10.1038/s41585-019-0184-4
32. Denzinger S, Burger M, Walter B, et al. Clinically relevant reduction in risk of recurrence of superficial bladder cancer using 5-aminolevulinic acid-induced fluorescence diagnosis: 8-year results of prospective randomized study. Urology. 2007;69(4):675-679. doi:10.1016/j.urology.2006.12.023
33. Fradet Y, Grossman HB, Gomella L, et al. A comparison of hexaminolevulinate fluorescence cystoscopy and white light cystoscopy for the detection of carcinoma in situ in patients with bladder cancer: a phase III, multicenter study. J Urol. 2007;178(1):68-73. doi:10.1016/j.juro.2007.03.028
34. Tadrist A, Gondran-Tellier B, McManus R, et al. Primary complete transurethral resection of bladder tumor using photodynamic diagnosis for high-risk nonmuscle invasive bladder cancer: is a restaging photodynamic transurethral resection really necessary? J Endourol. 2021;35(7):1042-1046. doi:10.1089/end.2020.1107
35. Klempfner H, Anderson P. Comparison of staging MRI to re‐resection for localised bladder cancer: narrative review. BJUI Compass. 2024;5(7):765-775. doi:10.1002/bco2.365
36. Khwaja SA, Caglic I, Shaida N, Colquhoun AJ, Turner W, Barrett T. Evaluation of magnetic resonance imaging for bladder cancer detection following transurethral resection of bladder tumour (TURBT). Abdom Radiol (NY). 2024;49(7):2340-2348. doi:10.1007/s00261-024-04235-6
37. Shkolyar E, Zhou SR, Carlson CJ, et al. Optimizing cystoscopy and TURBT: enhanced imaging and artificial intelligence. Nat Rev Urol. 2025;22(1):46-54. doi:10.1038/s41585-024-00904-9
38. Ali N, Bolenz C, Todenhöfer T, et al. Deep learning-based classification of blue light cystoscopy imaging during transurethral resection of bladder tumors. Sci Rep. 2021;11(1):11629. doi:10.1038/s41598-021-91081-x
39. Abbas S, Shafik R, Soomro N, Heer R, Adhikari K. AI predicting recurrence in non-muscle-invasive bladder cancer: systematic review with study strengths and weaknesses. Front Oncol. 2025;14:1509362. doi:10.3389/fonc.2024.1509362
40. Huang H, Huang Y, Kaggie JD, et al. Multiparametric MRI-based deep learning radiomics model for assessing 5-year recurrence risk in non-muscle invasive bladder cancer. J Magn Reson Imaging. 2025;61(3):1442-1456. doi:10.1002/jmri.29574
41. Guancial EA, Roussel B, Bergsma DP, et al. Bladder cancer in the elderly patient: challenges and solutions. Clin Interv Aging. 2015;10:939-949. doi:10.2147/CIA.S74322
42. Tognoni P, Simonato A, Robutti N, et al. Preoperative risk factors for postoperative delirium (POD) after urological surgery in the elderly. Arch Gerontol Geriatr. 2011;52(3):e166-e169. doi:10.1016/j.archger.2010.10.021
43. Lemiński A, Kaczmarek K, Gołąb A, Kotfis K, Skonieczna-Żydecka K, Słojewski M. Increased one-year mortality among elderly patients after radical cystectomy for muscle-invasive bladder cancer: a retrospective, observational comparative study. Clin Interv Aging. 2022;17:255-263. doi:10.2147/CIA.S352890
44. Iwamura H, Hatakeyama S, Momota M, et al. Relationship of frailty with treatment modality selection in patients with muscle-invasive bladder cancer (FRART-BC study). Transl Androl Urol. 2021;10(3):1143-1151. doi:10.21037/tau-20-1351
45. Black PC, Efstathiou J. Setting the stage for bladder preservation. Urol Oncol. 2021;39(4):209-212. doi:10.1016/j.urolonc.2020.09.001
46. Chen RC, Shipley WU, Efstathiou JA, Zietman AL. Trimodality bladder preservation therapy for muscle-invasive bladder cancer. J Natl Compr Canc Netw. 2013;11(8):952-960. doi:10.6004/jnccn.2013.0116
47. Efstathiou JA, Spiegel DY, Shipley WU, et al. Long-term outcomes of selective bladder preservation by combined-modality therapy for invasive bladder cancer: the MGH experience. Eur Urol. 2012;61(4):705-711. doi:10.1016/j.eururo.2011.11.010
Article Information
Published: 3/31/2025.
Conflict of Interest Disclosures: The authors have nothing to disclose.
Funding/Support: No funding or material support was received.
Author Contributions: All authors contributed equally to the research and writing of this article.
Data Availability Statement: No new data were generated for this article. All data cited are publicly available.