Introduction
Complicated urinary tract infections (UTIs) are a substantial health burden in the United States, particularly among patients with complex medical profiles or comorbid conditions. These infections, often driven by multidrug-resistant pathogens, are challenging to diagnose and manage effectively, increasing health care costs and straining clinical resources.1-3 Polymerase chain reaction (PCR)–based diagnostic tests have emerged as a promising alternative to conventional urine culture and sensitivity methods, offering faster pathogen identification and simultaneous detection of resistance genes, thereby enabling more informed selection of antibiotics.1 Despite these advantages, obtaining Medicare reimbursement through the Molecular Diagnostic Services Program (MolDX) for PCR testing for the diagnosis of complicated UTI remains a complex process that requires robust evidence.4 This review aimed to assess the analytical validity and clinical utility and validity of PCR-based testing compared with culture and sensitivity methods and to evaluate the alignment of current evidence with MolDX criteria for Medicare coverage.
Key Points
- Clinical validity and enhanced diagnostic accuracy. Polymerase chain reaction–based diagnostic testing demonstrates superior sensitivity, specificity, and rapidity compared with traditional culture and sensitivity methods.
- Clinical utility and economic benefits. Evidence highlights improved patient management, cost-effectiveness, and better health outcomes with PCR-based diagnostics.
- Population-specific impact. Data confirm effectiveness in older female adults with comorbidities as well as in patients with polymicrobial infections.
- Regulatory compliance and validation. This review identified 1 PCR panel that meets MolDX requirements and is backed by peer-reviewed evidence.
Abbreviations
MolDXMolecular Diagnostic Services Program
PCR polymerase chain reaction
UTI urinary tract infection
The economic impact of complicated UTIs on the US health care system is significant: Simmering et al3 demonstrated a steady rise in UTI-related hospitalizations from 1998 to 2011, with annual costs exceeding $2.8 billion. This financial burden is further compounded by the protracted hospital stays required for patients with complicated UTIs, which often arise from limitations in timely and accurate pathogen identification using standard urine culture methods.1,3 Delays in diagnosis can necessitate the use of broad-spectrum antibiotics, ultimately worsening patients’ antimicrobial resistance and escalating costs as a result of extended care and frequent readmissions.2,3
In recent studies, PCR-based diagnostics have had a notable impact on improving clinical and economic outcomes in complicated UTI management. For instance, Ko et al5 reported that PCR, when combined with antibiotic susceptibility testing, significantly reduced health care costs compared with culture and sensitivity testing. Their study demonstrated that patients undergoing PCR diagnostics experienced shorter hospital stays and fewer follow-up visits, directly affecting cost-effectiveness and patient satisfaction.5 Rapid PCR-based diagnostics enable faster, pathogen-specific treatment, which could streamline patient care pathways and reduce reliance on broad-spectrum antibiotics.1 In addition, Korman et al6 reported that multiplex PCR testing combined with pooled antibiotic susceptibility testing resulted in a reduction in antibiotic resistance in patients with symptomatic complicated UTI in urology settings, indicating that such testing is valuable in the management of these types of infections. These findings emphasize PCR’s potential to support responsible antibiotic stewardship without exacerbating resistance concerns, which addresses a key challenge in complicated UTI management.6
Several countries, including the United Kingdom, Germany, and Japan, have recognized the clinical benefits of PCR diagnostics for complicated UTIs in specific contexts, particularly for rapidly identifying pathogens and resistance genes in complex or resistant infections. Reimbursement for PCR diagnostics in these countries tends to be selective, favoring cases in which rapid and precise results are critical for patient outcomes, such as in severe,
recurrent, or treatment-resistant complicated UTI.1,7 The United States has been slower in adopting the reimbursement of large PCR panels for complicated UTI, partly because of the rigorous reimbursement criteria set by MolDX. The program requires comprehensive evidence of a diagnostic tool’s clinical validity and utility for Medicare reimbursement. It specifically outlines that a PCR panel must demonstrate high sensitivity, specificity, and reproducibility as well as a proven impact on clinical outcomes, such as reduced symptom duration, fewer hospitalizations, and cost-effectiveness (Table 1).4
This review examines the current body of literature on PCR-based diagnostics for complicated UTI and discusses the extent to which existing studies meet MolDX requirements for Medicare reimbursement—specifically, clinical validity and clinical utility criteria (Table 1). Through an analysis of real-world evidence, clinical trials, systematic reviews, and meta-analyses, this review identifies strengths and gaps in the current evidence base and highlights areas where further research is needed to fully meet MolDX’s standards for coverage.
Real-World Evidence
Real-World Evidence Studies Summary
Real-world evidence studies derived from real-world data analysis provide critical insights into the usefulness of PCR diagnostics over culture and sensitivity testing in managing complicated UTI, especially regarding clinical decision-making and patient outcomes. These studies are essential for evaluating the effectiveness of PCR in routine clinical practice, a key MolDX criterion for Medicare reimbursement approval. In this review, we analyzed the scope, objectives, and findings of relevant real-world evidence studies, assessing their alignment with MolDX’s analytical and clinical utility standards.

Ko et al5 provided compelling real-world evidence supporting the adoption of a novel diagnostic tool that combines molecular testing with pooled antibiotic susceptibility testing for managing complicated UTI. The study demonstrated statistically significant reductions in overall health care costs compared with traditional culture and sensitivity testing; the mean total cost in the PCR-guided cohort was $629.55 vs $1131.39 in the culture and sensitivity group. These figures represent a statistically significant cost difference of $501.84 (95% CI, 192.25-709.18; P = .004). The cost-effectiveness of molecular diagnostic testing is further emphasized by its ability to reduce health care expenditures across care settings, including outpatient, inpatient, and long-term care facilities.5 These outcomes were achieved through earlier, more targeted therapeutic interventions, minimizing clinical reliance on broad-spectrum antibiotics and reducing the risk of complications. The diagnostic process using PCR also improved clinical management by enabling rapid pathogen identification and facilitating tailored treatment decisions.
Festa et al8 evaluated the diagnostic performance of multiplex PCR in detecting viable microorganisms in urine samples from patients with suspected UTIs. The study compared microorganism identification using multiplex PCR with standard urine culture. Multiplex PCR identified 395 organisms, substantially outperforming standard urine culture, which detected only 108 organisms (27.3% of the total identified by PCR; P < .0001). Among nonfastidious bacteria, multiplex PCR detected 260 organisms, whereas standard urine culture identified 108 (41.5% of the total detected by PCR; P < .0001). Common pathogens that standard urine culture frequently missed but multiplex PCR identified included Escherichia coli (detected in 72.5% of cases by standard urine culture), Klebsiella pneumoniae (66.7%), Enterococcus faecalis (34.6%), and Enterococcus faecium (0%). Standard urine culture failed to detect any of the 135 fastidious bacteria identified by multiplex PCR (P < .0001). Fastidious bacteria are microorganisms that require specific growth conditions and may be difficult to cultivate using standard culture methods. These findings highlight the superior sensitivity and specificity of multiplex PCR in identifying viable pathogens, underscoring its analytical validity and potential to enhance targeted antimicrobial therapy in UTI management.8
Kapoor et al9 conducted a comprehensive study comparing the diagnostic accuracy of PCR testing and standard urine culture in managing complicated UTI. Their study analyzed data from 3395 patients in 2022 who underwent simultaneous PCR and standard urine culture testing. Polymerase chain reaction testing demonstrated a significantly higher pathogen detection rate, identifying 36.4% more organisms than did standard urine culture testing. Moreover, PCR was 20 times more effective in detecting organisms responsible for polymicrobial infections and nearly 36 times more effective in identifying fastidious organisms. Concordance analysis revealed that PCR identified 90.6% of the organisms found by standard urine culture, but standard urine culture detected only 40.7% of the organisms identified by PCR. Notably, 62.4% of the organisms detected by PCR were missed by standard urine culture testing, while standard urine culture testing identified 9.4% of organisms not detected by PCR, with all differences being statistically significant (P < .05). These findings strongly support the use of PCR to enhance diagnostic accuracy, reduce false negatives, and improve treatment strategies for patients with complicated UTI involving complex or hard-to-culture pathogens.9
Hao et al10 focused on the essential role of PCR, especially larger PCR panel sizes, in accurately identifying polymicrobial infections and fastidious organisms in complicated UTI. This study underscored that PCR’s broad detection spectrum is particularly valuable for patients with complicated UTI, whose infections may involve multiple or atypical pathogens that evade detection by standard urine culture. Polymerase chain reaction testing significantly outperforms standard urine culture in detecting pathogens in complicated UTI, particularly polymicrobial infections and fastidious organisms. Line-item concordance analysis further demonstrated that PCR detected 90.2% of organisms identified by standard urine culture, while standard urine culture detected only 31.9% of organisms found by PCR (P < .01). In addition, polymerase chain reaction testing detected more polymicrobial infections and fastidious organisms than standard urine culture (polymicrobial infections, 46.2% vs 3.6%; fastidious organisms, 31.3% vs 0.7%; both P < .01). Organism detection increased with expansion in PCR panel size from 5 to 25 organisms (P < .01). The study data show that PCR testing significantly improves the overall positivity rate and detection of individual organisms, including polymicrobial infections and fastidious organisms, with these benefits being most pronounced when a PCR panel size of 25 or greater is used.10 The authors found that, unlike standard urine culture methods, which are limited to identifying culturable pathogens, PCR provides a more comprehensive diagnostic profile by detecting organisms that standard urine culture methods often fail to isolate,10 thereby supporting analytical and clinical diagnostic validity requirements.
Haley et al11 conducted a comparative study to assess the clinical impact of multiplex PCR combined with pooled antibiotic susceptibility testing vs standard urine culture in managing complicated and recurrent UTI. The study included 577 symptomatic adults (207 men, 370 women) treated between March 2022 and May 2023. Patients diagnosed using multiplex PCR/pooled antibiotic susceptibility testing (n = 252) experienced significantly reduced rates of empirical antibiotic use (28.7% vs 66.7%) compared with patients in the standard urine culture group (n = 146). In addition, the multiplex PCR/pooled antibiotic susceptibility testing arm demonstrated lower composite negative outcomes (34.5% vs 46.6%; P = .018), including fewer UTI-related medical visits and hospitalizations (P < .05). Patients aged 60 years and older in the multiplex PCR/pooled antibiotic susceptibility testing group reported a statistically significant reduction in UTI symptom recurrence (P < .05). The turnaround time for multiplex PCR/pooled antibiotic susceptibility testing results was approximately half that of standard urine culture (P < .0001), enabling faster clinical decision-making. These findings highlight the advantages of multiplex PCR/pooled antibiotic susceptibility testing in reducing unnecessary empirical treatments, improving clinical outcomes, and minimizing symptom recurrence in patients with complicated UTI.11
In another study by Korman et al,12 the authors evaluated the impact of a molecular and phenotypic diagnostic approach using multiplex PCR/pooled antibiotic susceptibility testing in managing complicated UTI, focusing on patients with non–E coli or polymicrobial infections. Among 264 patients with suspected complicated UTI, 146 (55.3%) had exclusively non–E coli infections, and 190 (72%) presented with polymicrobial infections; treated patients with non–E coli infections exhibited significantly greater symptom reduction on day 14 compared with untreated patients (3.18 vs 1.64; P = .006). Similarly, for polymicrobial infections, treated patients experienced greater symptom reduction (3.52 vs 1.41; P = .002) and higher clinical cure rates (58.7% vs 36.4%; P = .049). These findings emphasize the clinical utility of multiplex PCR/pooled antibiotic susceptibility testing in guiding targeted treatments, reducing symptom burden, and achieving improved clinical outcomes for patients with complicated UTI whose complex cases are not adequately addressed by standard urine culture methods.12
Elia et al13 investigated the clinical utility of urinary PCR testing in managing complicated UTI by analyzing how PCR results influenced therapeutic decisions compared with standard urine culture testing. The study included 96 surveys completed by 21 clinicians treating patients with symptomatic complicated UTI. Polymerase chain reaction testing led to modification in the antibiotic regimen for 61.5% (59/96) of patients. Notably, in 52.5% (31/59) of these patients, PCR results prompted treatment changes that standard urine culture alone would not have suggested, highlighting PCR testing’s potential to enhance antibiotic stewardship. This study underscores PCR’s capacity to enable timely, pathogen-specific therapeutic adjustments, particularly in challenging complicated UTI cases, thereby facilitating improved clinical outcomes and more effective use of antibiotics.13
Wojno et al14 provided robust evidence supporting the diagnostic utility of multiplex PCR testing for UTI. In a retrospective study involving 582 symptomatic older patients (mean age, 77 years), PCR testing demonstrated superior sensitivity by detecting uropathogens in 56% (326/582) of patients compared with 37% (217/582) of patients tested using standard urine culture. Notably, PCR identified pathogens in 22% (130/582) of patients for whom standard urine culture results were negative. Polymicrobial infections were reported more comprehensively by PCR testing (166 patients) than by standard urine culture (39 patients), with PCR revealing 67 instances of polymicrobial infections undetected by standard urine culture. The agreement between PCR and standard urine culture was 90% for positive cases, exceeding the noninferiority threshold of 85% (95% CI, 85.7%-93.6%). These findings underscore PCR’s enhanced diagnostic capabilities, particularly in identifying clinically relevant pathogens that standard urine culture may miss. The study highlights PCR’s analytical validity, reproducibility, and potential to improve patient outcomes through faster and more accurate pathogen detection. This study positioned PCR as a valuable tool in optimizing UTI diagnosis and management, especially in patients with complex infections.14
Analysis of Real-World Evidence and MolDX Criteria Alignment
The reviewed real-world evidence studies collectively demonstrate PCR’s potential to meet some key MolDX requirements for Medicare reimbursement. Festa et al,8 Kapoor et al,9 Hao et al,10 and Wojno et al14 support the analytical validity criterion, showing PCR’s advantages in relation to diagnostic analytical validity, accuracy, and pathogen coverage. Haley et al,11 Korman et al,12 and Elia et al13 partially address clinical correlation and demonstrate improved disease management and health outcomes with PCR-informed treatments. In addition, Ko et al5 demonstrated that molecular diagnostic testing offers significant cost savings compared with traditional culture and sensitivity methods while meeting MolDX’s clinical validity and utility standards.
Despite the extensive findings from these real-world evidence studies that support PCR’s analytical validity and potential clinical utility in complicated UTI management, a critical gap remains in the evidence base: direct prospective comparisons of patient outcomes between patients prescribed PCR-guided treatments vs patients treated based on conventional culture and sensitivity methods. Although these studies demonstrate PCR’s advantages in diagnostic accuracy, cost-effectiveness, and reduced turnaround time, they fall short of documenting improvements in clinical outcomes such as symptom resolution for patients prescribed PCR-guided treatment compared with patients treated based on culture and sensitivity results. The current body of real-world evidence does not therefore fully satisfy MolDX’s reimbursement criteria, which require clear, demonstrated improvement in patient outcomes attributable to the use of PCR over standard culture and sensitivity diagnostics.
Systematic Review and Meta-Analysis
Systematic Review and Meta-Analysis Summary
The role of molecular diagnostics—specifically, PCR testing—in diagnosing UTIs has gained considerable attention because of the technology’s potential for improving diagnostic speed and accuracy compared with culture and sensitivity. Systematic reviews and meta-analyses provide valuable insights into the overall performance and clinical utility of PCR-based methods for complicated UTI diagnosis. Three recent studies analyzed the diagnostic accuracy, clinical impact, and utility of PCR vs standard urine culture, summarizing current evidence and addressing the generalizability of molecular diagnostics in UTI management.
Robledo et al15 conducted a systematic review and meta-analysis to evaluate the accuracy of molecular diagnostics in patients with confirmed, culture-based UTI diagnoses. This study found that PCR-based diagnostics demonstrated high sensitivity and specificity; the overall sensitivity was 0.80 (95% CI, 0.73-0.86), and specificity was 0.83 (95% CI, 0.52-0.95).15 Diagnosis by standard urine culture, which requires more than 24 hours for results, often necessitates the use of empiric antibiotics without pathogen confirmation. In contrast, PCR delivers faster, more reliable results and serves as an effective screening tool. It is simple, cost-efficient, and suitable for point-of-care use, providing pathogen detection and simultaneous antimicrobial susceptibility results within hours.15 This rapid turnaround minimizes unnecessary empiric antibiotic use and supports precise, pathogen-specific therapy. The study underscored the advantage of PCR in identifying fastidious and polymicrobial infections, which are often missed by standard urine culture methods, highlighting the technology’s potential as a more comprehensive diagnostic tool for complicated UTI management.
Szlachta-McGinn et al16 focused on the diagnostic accuracy and therapeutic impact of molecular diagnostics, comparing PCR and standard urine culture methods in diagnosing UTI. Their meta-analysis demonstrated PCR’s higher pathogen detection rates, especially for mixed or less common infections often missed by standard urine culture.16 By identifying a wider array of pathogens, PCR can provide clinicians with more actionable data for targeted therapy, thereby potentially improving treatment outcomes.
Zhao et al17 compared PCR and next-generation sequencing (NGS) with standard urine culture tests in diagnosing UTIs. Their meta-analysis concluded that PCR had superior diagnostic performance, particularly for patients with polymicrobial infections or pathogens that are difficult to culture.17 These molecular methods effectively detected a broad spectrum of gram-positive bacteria, such as Staphylococcus saprophyticus, Streptococcus, and Corynebacterium urealyticum, as well as gram-negative bacteria, such as E coli, Pseudomonas, and Proteus mirabilis. In UTIs, which are frequently caused by E coli and S saprophyticus, these techniques surpass standard urine culture testing by offering comprehensive evaluations of the urinary microbiome.17 The study’s findings underscore PCR’s potential to enhance the accuracy and speed of UTI diagnosis.
Analysis of Systematic Review and Meta-Analysis Evidence and MolDX Criteria Alignment
These systematic reviews and meta-analyses collectively confirm the diagnostic superiority of PCR over culture and sensitivity in complicated UTI detection, particularly for identifying complex and mixed infections. They primarily focused on analytical performance metrics, however, and lack comprehensive data on patient outcomes and clinical benefits associated with PCR-guided treatment. As such, although these reviews support PCR’s potential utility, they do not fully satisfy MolDX criteria, which emphasize improvements in patient management and outcomes as essential for reimbursement approval.
Clinical Trials
Clinical Trial Summary
The clinical trial data published by Spangler et al18,19 reinforced the comparative utility of PCR diagnostics vs traditional culture and sensitivity testing for managing complicated UTI. These trial data were reported in a 2-part publication, marking the first rigorous investigation of its kind. Part 1 explored clinical outcomes, health care investigator satisfaction levels, and diagnostic turnaround times specifically for patients with complicated UTI. Part 2 examined clinical validation, concordance, and antibiotic selection. Together, these publications provide a comprehensive evaluation of the clinical utility of PCR, highlighting its substantial advantages in real-world settings. The trial involved 773 adult patients with confirmed symptomatic complicated UTI and assessed both the clinical and operational impacts of PCR testing compared with the conventional culture and sensitivity approach.18,19
The primary goal of Spangler et al18,19 was to explore whether PCR testing could improve clinical outcomes by enabling faster, more accurate pathogen identification and more efficient antibiotic selection, ultimately reducing the time to appropriate therapy. In addition, the study evaluated investigator satisfaction and documented turnaround times as key operational metrics to compare PCR’s effectiveness in a clinical workflow with that of culture and sensitivity.18,19
The trial highlighted the statistically significant advantages of PCR-based diagnostics over culture and sensitivity testing for managing complicated UTI. Polymerase chain reaction testing demonstrated a notably shorter turnaround time (49.68 hours vs 104.4 hours; P < .001), enabling faster clinical decision-making. Investigators also reported higher satisfaction scores for PCR (PCR mean [SD] satisfaction score, 23.95 [1.96] vs culture and sensitivity mean [SD] satisfaction score, 20.64 [4.12]; P < .001), emphasizing its usefulness in optimizing treatment strategies. Treatments guided by PCR results yielded better clinical outcomes than did treatments based on culture and sensitivity testing (88.08% vs 78.11%; P = .011). In addition, PCR-guided treatment favored the use of oral antibiotics, which achieved 87.15% favorable clinical outcomes, significantly higher than the 77.37% favorable clinical outcomes observed in the culture and sensitivity group (P = .018). The study also showed strong agreement between PCR and culture and sensitivity testing in diagnosing positive cases (88.06% at the end of the study) but noted discordance in negative cases (62.91%) mainly because of the potential limitation of culture and sensitivity in identifying uropathogens. Polymerase chain reaction diagnostics identified 102 complicated UTI cases missed by culture and sensitivity testing, while culture and sensitivity testing detected only 14 cases that PCR did not. Of the patients in the study with discordant results, the individuals treated with PCR-guided medication exhibited better clinical outcomes compared with patients diagnosed by culture and sensitivity testing (77.45% vs 71.42%).18,19
Analysis of Clinical Trials Evidence and MolDX Criteria Alignment
Although the findings from Spangler et al18,19 contribute robust evidence in support of PCR’s validity and utility, the study lacks data specifically linking these benefits to cost savings and sustained health improvements in a Medicare-focused cohort.
Analysis of the Body of Evidence
The comprehensive evidence provided by real-world studies, clinical trials, systematic reviews, and meta-analyses demonstrates the potential of PCR-based diagnostics to meet MolDX’s stringent criteria for Medicare coverage in managing complicated UTI (Table 2). Regarding clinical validity, PCR testing has shown high sensitivity, specificity, and accuracy in pathogen detection, including fastidious organisms and polymicrobial infections often missed by traditional culture and sensitivity methods.5,8-17 These findings align with MolDX’s emphasis on providing reliable and actionable pathogen identification, providing clinicians with precise diagnostic data for effective treatment decision-making.
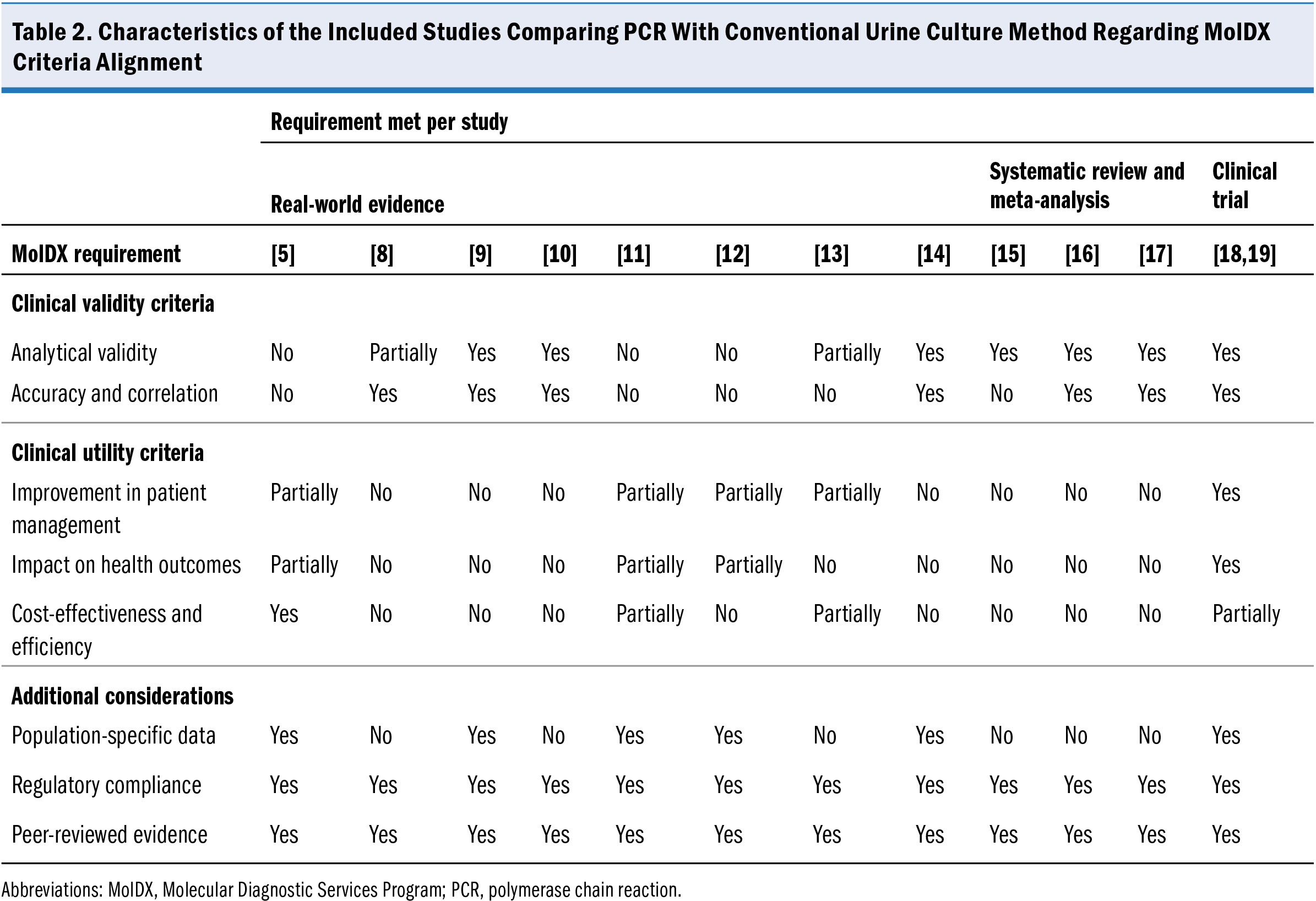
For MolDX’s clinical utility criteria, which mandate evidence of improved patient management, cost-effectiveness, and health outcomes, the implementation of PCR testing has shown substantial advantages. Ko et al5 demonstrated that integrating PCR with antibiotic susceptibility testing helped curb health care costs by minimizing unnecessary broad-spectrum antibiotic use and limiting the potential for infection-related complications. This approach addresses a critical clinical need for precision in treatment selection, one that directly targets causative pathogens, reduces infection progression, and helps break the cycle of recurrent infections.
Spangler et al18,19 furthermore highlighted an essential need for more rapid diagnostics in clinical settings, with PCR providing results within hours as opposed to the prolonged 2-day to 3-day delay common with traditional culture and sensitivity testing. This accelerated diagnostic feedback enables clinicians to adjust treatment protocols quickly, leading to faster symptom resolution; less patient discomfort; and a reduced risk for hospital admissions, readmissions, and extended stays. Enhanced decision-making speed directly supports patient satisfaction and symptom control, which are crucial outcomes in complicated UTI care. Spangler et al18,19 also reported higher clinician satisfaction with PCR testing because of its efficiency in streamlining patient care and reducing reliance on empirical antibiotic choices, results that align closely with MolDX’s focus on improving patient care management. The clinical utility of PCR was demonstrated in the improvement of therapeutic clinical outcomes and the enabling of expedited, patient-specific management of complicated UTI for optimal patient care. These findings highlight a shift toward a more responsive, tailored approach to complicated UTI management, meeting critical clinical needs for superior patient outcomes, including symptom reduction and optimized antibiotic stewardship.
Conclusion
The collective evidence—spanning real-world data, clinical trials, systematic reviews, and meta-analyses—strongly supports the clinical validity of PCR-based testing for patients with complicated UTI. Studies have consistently demonstrated PCR testing’s superior diagnostic accuracy and reliability compared with traditional culture and sensitivity tests, and its robust analytical validity and clinical utility have been demonstrated across diverse patient populations.
The clinical validity and utility of the PCR syndromic panel evaluated by Spangler et al18,19 and the cost-effectiveness findings of Ko et al,5 which apply broadly to molecular testing with pooled antibiotic susceptibility testing, satisfy all MolDX criteria, substantiating the case for reimbursement approval. Spangler et al18,19 supported the panel’s clinical validity by demonstrating high sensitivity, specificity, accuracy, and rapid diagnostic turnaround times, which allow for more precise and timely treatment decisions while reducing clinical reliance on empirical antibiotics. In addition, the clinical utility of Spangler et al’s PCR syndromic panel was confirmed by the way it improved patient outcomes, including reducing symptoms, introducing a break in the recurrent infection cycle, and providing higher clinician satisfaction, thereby emphasizing the PCR panel’s alignment with MolDX’s focus on enhanced patient care and treatment outcomes. Complementing these results, Ko et al5 provided compelling evidence for PCR testing’s cost-effectiveness, demonstrating reduced infection severity and health care expenditures through earlier targeted interventions that prevented complications and improved resource utilization. These findings reinforce Spangler et al’s PCR panel’s capacity to meet MolDX’s analytical, clinical, and economic criteria, positioning it as an essential, transformative diagnostic tool for modern complicated UTI management.
References
1. Xu R, Deebel N, Casals R, Dutta R, Mirzazadeh M. A new gold rush: a review of current and developing diagnostic tools for urinary tract infections. Diagnostics (Basel). 2021;11(3):479. doi:10.3390/diagnostics11030479
2. Laudisio A, Marinosci F, Fontana D, et al. The burden of comorbidity is associated with symptomatic polymicrobial urinary tract infection among institutionalized elderly. Aging Clin Exp Res. 2015;27(6):805-812. doi:10.1007/s40520-015-0364-x
3. Simmering JE, Tang F, Cavanaugh JE, Polgreen LA, Polgreen PM. The increase in hospitalizations for urinary tract infections and the associated costs in the United States, 1998-2011. Open Forum Infect Dis. 2017;4(1):ofw281. doi:10.1093/ofid/ofw281
4. MolDX: Molecular diagnostic tests (MDT) (L36807). Centers for Medicare & Medicaid Services. Updated April 21, 2023. Accessed November 11, 2024. https://www.cms.gov/medicare-coverage-database/view/lcd.aspx?lcdId=36807
5. Ko DSC, Lukacz ES, Juster IA, et al. Real-world evidence that a novel diagnostic combining molecular testing with pooled antibiotic susceptibility testing is associated with reduced infection severity and lower cost compared with standard urine culture in patients with complicated or persistently recurrent urinary tract infections. JU Open Plus. 2023;1(5):e00021. doi:10.1097/JU9.0000000000000025
6. Korman HJ, Mathur M, Luke N, et al. Multiplex polymerase chain reaction/pooled antibiotic susceptibility testing was not associated with increased antibiotic resistance in management of complicated urinary tract infections. Infect Drug Resist. 2023;16:2841-2848. doi:10.2147/IDR.S406745
7. Messacar K, Parker SK, Todd JK, Dominguez SR. Implementation of rapid molecular infectious disease diagnostics: the role of diagnostic and antimicrobial stewardship. J Clin Microbiol. 2017;55(3):715-723. doi:10.1128/JCM.02264-16
8. Festa RA, Luke N, Mathur M, et al. A test combining multiplex-PCR with pooled antibiotic susceptibility testing has high correlation with expanded urine culture for detection of live bacteria in urine samples of suspected UTI patients. Diagn Microbiol Infect Dis. 2023;107(2):116015. doi:10.1016/j.diagmicrobio.2023.116015
9. Kapoor DA, Holton MR, Hafron J, Aljundi R, Zwaans B, Hollander M. Comparison of polymerase chain reaction and urine culture in the evaluation of patients with complex urinary tract infections. Biology (Basel). 2024;13(4):257. doi:10.3390/biology13040257
10. Hao X, Cognetti M, Patel C, et al. The essential role of PCR and PCR panel size in comparison with urine culture in identification of polymicrobial and fastidious organisms in patients with complicated urinary tract infections. Int J Mol Sci. 2023;24(18):14269. doi:10.3390/ijms241814269
11. Haley E, Luke N, Korman H, et al. Improving patient outcomes while reducing empirical treatment with multiplex-polymerase-chain-reaction/pooled-antibiotic-susceptibility-testing assay for complicated and recurrent urinary tract infections. Diagnostics (Basel). 2023;13(19):3060. doi:10.3390/diagnostics13193060
12. Korman HJ, Baunoch D, Luke N, et al. A diagnostic test combining molecular testing with phenotypic pooled antibiotic susceptibility improved the clinical outcomes of patients with non-E. coli or polymicrobial complicated urinary tract infections. Res Rep Urol. 2023;15:141-147. doi:10.2147/RRU.S404260
13. Elia J, Hafron J, Holton M, Ervin C, Hollander MB, Kapoor DA. The impact of polymerase chain reaction urine testing on clinical decision-making in the management of complex urinary tract infections. Int J Mol Sci. 2024;25(12):6616. doi:10.3390/ijms25126616
14. Wojno KJ, Baunoch D, Luke N, et al. Multiplex PCR-based urinary tract infection (UTI) analysis compared to traditional urine culture in identifying significant pathogens in symptomatic patients. Urology. 2020;136:119-126. doi:10.1016/j.urology.2019.10.018
15. Robledo XG, Arcila KVO, Riascos SHM, García-Perdomo HA. Accuracy of molecular diagnostic techniques in patients with a confirmed urine culture: a systematic review and meta-analysis. Can Urol Assoc J. 2022;16(9):E484-E489. doi:10.5489/cuaj.7677
16. Szlachta-McGinn A, Douglass KM, Chung UYR, Jackson NJ, Nickel JC, Ackerman AL. Molecular diagnostic methods versus conventional urine culture for diagnosis and treatment of urinary tract infection: a systematic review and meta-analysis. Eur Urol Open Sci. 2022;44:113-124. doi:10.1016/j.euros.2022.08.009
17. Zhao M, Qi S, Sun Y, Zheng X. Comparison of polymerase chain reaction and next-generation sequencing with conventional urine culture for the diagnosis of urinary tract infections: a meta-analysis. Open Med (Wars). 2024;19(1):20240921. doi:10.1515/med-2024-0921
18. Spangler FL, Williams C, Aberger ME, et al. Clinical utility of PCR compared to conventional culture and sensitivity testing for the management of complicated urinary tract infections in adults: part I. Assessment of clinical outcomes, investigator satisfaction scores, and turnaround times. Diagn Microbiol Infect Dis. 2025;111(1):116601. doi:10.1016/j.diagmicrobio.2024.116601
19. Spangler FL, Williams C, Aberger ME, et al. Clinical utility of PCR compared to conventional culture and sensitivity testing for the management of complicated urinary tract infections in adults: part II. Evaluation of diagnostic concordance, discordant results, and antimicrobial selection efficacy. Diagn Microbiol Infect Dis. 2024;111(3):116646. doi:10.1016/j.diagmicrobio.2024.116646
Article Information
Published: 3/31/2025.
Conflict of Interest Disclosures: The authors acknowledge that the sponsor’s funding supported the evaluation of the DocLab PCR panel and its alignment with MolDX coverage criteria. Although this review identified the DocLab PCR panel as meeting many of the MolDX requirements for reimbursement, all analyses and interpretations presented in this manuscript were conducted independently and are based on an objective synthesis of the available evidence. The authors declare no other conflicts of interest related to this article.
Funding/Support: DocLab provided funding for the PCR panel assessed in this systematic review.
Author Contributions: Moustafa Kardjadj designed and led the study. Michael E. Aberger and Bradley A. Wilson served as co-first authors, contributing equally to the preparation and writing of the manuscript. All authors participated in data analysis and manuscript development.
Data Availability Statement: All data analyzed in this systematic review are derived from published studies. The datasets used or analyzed during the current study are publicly available from the respective sources cited in the article. No original data were collected or generated specifically for this review.