Introduction
Prostate cancer (PCa) is a leading cause of mortality and morbidity, both globally and in the United States (US), representing a substantial public health care burden.1-4 In the US, approximately 299 010 men were diagnosed in 2024, and 35 250 will die from the disease.2,3 US estimates suggest that most PCa cases are localized (70%) or regional (13%) at initial presentation, with an estimated 8% and 9% of patients presenting with distant disease and unknown stage, respectively.3,4
Abbreviations
1L first line
2L second line
EMR electronic medical record
FDA US Food and Drug Administration
HR hazard ratio
HRR homologous recombination repair
LOT line of therapy
mCRPC metastatic castration-resistant prostate cancer
mHSPC metastatic hormone-sensitive prostate cancer
NGS next-generation sequencing
NHA novel hormonal agent
OS overall survival
PARP poly(ADP-ribose) polymerase
PCa prostate cancer
PFS progression-free survival
PSA prostate-specific antigen
SDOH social determinants of health
TTNT time to next treatment
US United States
Metastatic castration-resistant prostate cancer (mCRPC) represents the most aggressive phenotype of the disease. While novel hormonal agents (NHAs) such as abiraterone and enzalutamide have demonstrated efficacy in delaying disease progression in multiple trials, a substantial proportion of patients will develop resistance to these agents over time.5-8 Across landmark clinical studies, including COU-AA-302 studying abiraterone and PREVAIL studying enzalutamide, median overall survival (OS) time frames were approximately 35 months.5-8 Treatment has also evolved for patients whose tumors are homologous recombination repair (HRR) or BRCA mutated. Specifically, poly(ADP-ribose) polymerase (PARP) inhibitors have emerged as a treatment option for certain patients with mCRPC, such as those with BRCA alterations who have received prior hormonal therapy and chemotherapy as well as those with a broader range of HRR mutations.9 As such, genetic testing in patients with mCRPC can help determine eligibility for PARP inhibitors, which can improve survival as monotherapy or as a component of combination therapy.9
Several studies of real-world clinical practice in mCRPC have been published. Caram et al10 utilized US Department of Veterans Affairs data to examine differences in survival, stratified by year of first CRPC treatment, initially finding survival times to have improved between 2010 and 2017. However, these benefits were mitigated when adjusting for markers of disease burden, suggesting that recent gains may be due to an earlier time to treatment initiation rather than improved treatment effectiveness. Malangone-Monaco and colleagues11 utilized US employer-sponsored claims data to improve upon understanding of treatment sequencing in patients with mCRPC, finding high patient attrition with successive lines of therapy, with only 47.8% and 21.8% of patients receiving second and third lines, respectively. Novel hormonal agents were found to be the most common first-line (1L), second-line (2L), and third-line treatment class; back-to-back use of alternate NHAs was found to be the most common treatment sequence.11 Lastly, Barata et al12 evaluated the impact of taxane-based chemotherapy and NHA use in the metastatic hormone-sensitive prostate cancer (mHSPC) setting on selection of 1L treatment regimen in the mCRPC setting. Their findings suggest that physicians initiate 1L treatment most commonly with an NHA, regardless of prior treatment.12
In the US, patients with PCa are cared for by urologists and/or oncologists; therefore, generating real-world evidence from both perspectives is important for enhanced understanding of real-world care. The goal of this study was to build upon published urology and oncology data by describing treatment patterns, sequencing, and outcomes for US patients, including Black/African Americans treated in a large network of urology practices, and with this information, discuss opportunities for improvement in patient care strategies.
Methods
This study was a retrospective analysis using the Specialty Networks Uro-oncology deidentified electronic medical records (EMR) database. This is a large US urology dataset consisting of records from EMR, practice management, and in-office dispensing and imaging systems from approximately 90 community urology practices with over 3200 providers. More than 1 million patients with PCa were identified. The database is representative of the US geography, payers, and relevant cohort ages and sex, and it aligns with the incidence and prevalence of US disease state comparisons as reported by the US Centers for Disease Control and Prevention.4,13
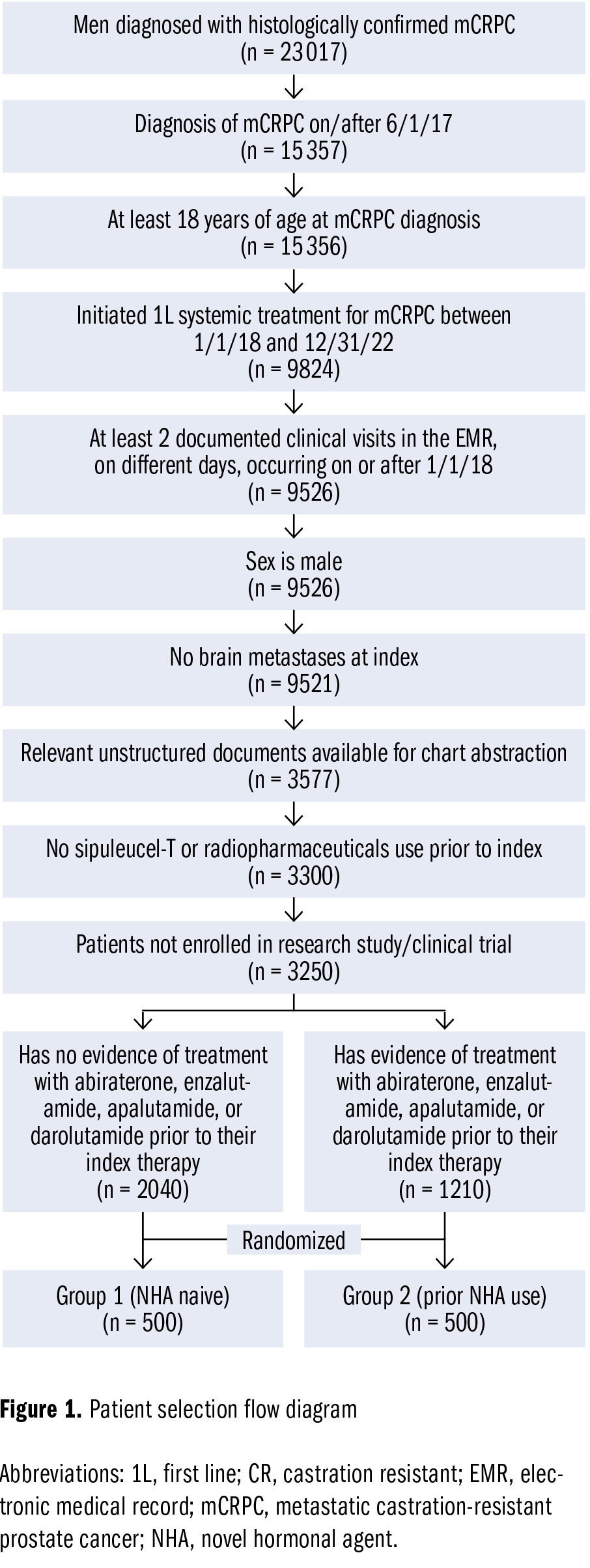
Adult patients with a diagnosis of mCRPC and who initiated 1L systemic treatment for mCRPC between January 1, 2018, and December 31, 2022, were identified using a random sampling–based methodology. Figure 1 shows a flow diagram of patient selection for the analysis. Patients were excluded if they had brain metastases at the time of 1L mCRPC treatment, lacked relevant unstructured documents for study data, had sipuleucel-T or radiopharmaceutical use prior to the index date, or were enrolled in a clinical trial involving an investigational product at any time during the study period. The index date was the start of 1L for mCRPC. This study contained 1000 patients comprised of 2 groups. Group 1 included patients who had not been treated with an NHA prior to 1L therapy for mCRPC (ie, NHA naive), and group 2 included patients who had been treated with an NHA prior to 1L therapy for mCRPC (ie, prior NHA use).
An algorithm to determine lines of therapy (LOTs) was developed based on prior literature and the clinical expertise of the study team. The patient’s first systemic treatment up to 14 days prior to the mCRPC diagnosis date was considered 1L mCRPC. Treatments added beyond 45 days after the initial regimen triggered an advancement in the LOT. A gap of up to 90 days for the same regimen was allowable before advancing the LOT. (See LOT rules in Supplement.) It is important to note that androgen-deprivation therapy use was not characterized as part of the LOT algorithm used for this study given that it is initiated early in patients’ disease course and continued as part of the treatment for metastatic disease.14
Primary objectives of the study were to describe patient clinical and demographic characteristics, utilization of 1L systemic regimens, and real-world outcomes for time to next treatment (TTNT) and OS. Secondary objectives included describing the proportion of patients who received an NHA for both pre-1L and 1L mCRPC therapy and the percentage of patients who received care by an oncologist (in addition to a urologist) for mCRPC. The study was exempt from institutional review board approval.
Statistical Analysis
Baseline patient characteristics for the overall population and for the 2 study groups were summarized using descriptive statistics. The χ2 or Fisher exact test, where applicable, was used to analyze categorical variables; the Welch 2-sample t test or Wilcoxon rank sum test, as appropriate, was used to analyze continuous variables between the 2 groups. Time-to-event outcomes were evaluated using Kaplan-Meier methods and Cox proportional hazards regression models adjusted for baseline patient characteristics, including cohort grouping (1 or 2), age, geography, insurance, race and ethnicity, initial stage at diagnosis, index year, Gleason score at initial PCa diagnosis, prostate-specific antigen (PSA) level at mCRPC diagnosis, 1L treatment type, and time from initial PCa to mCRPC diagnosis. Analysis was conducted using RStudio, version 1.2.5033, software (RStudio, Inc).
Results
Patient Demographics and Baseline Characteristics
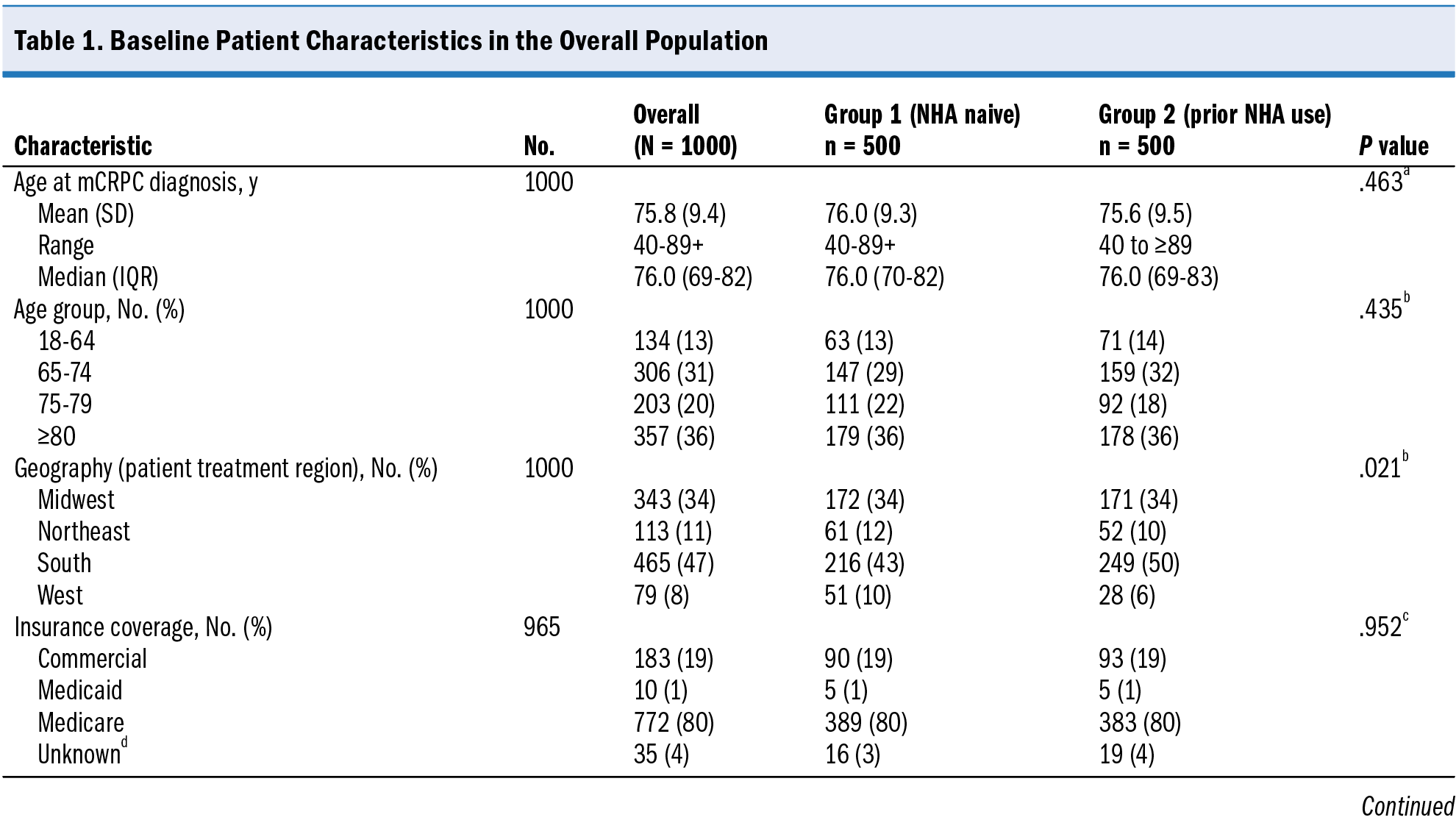
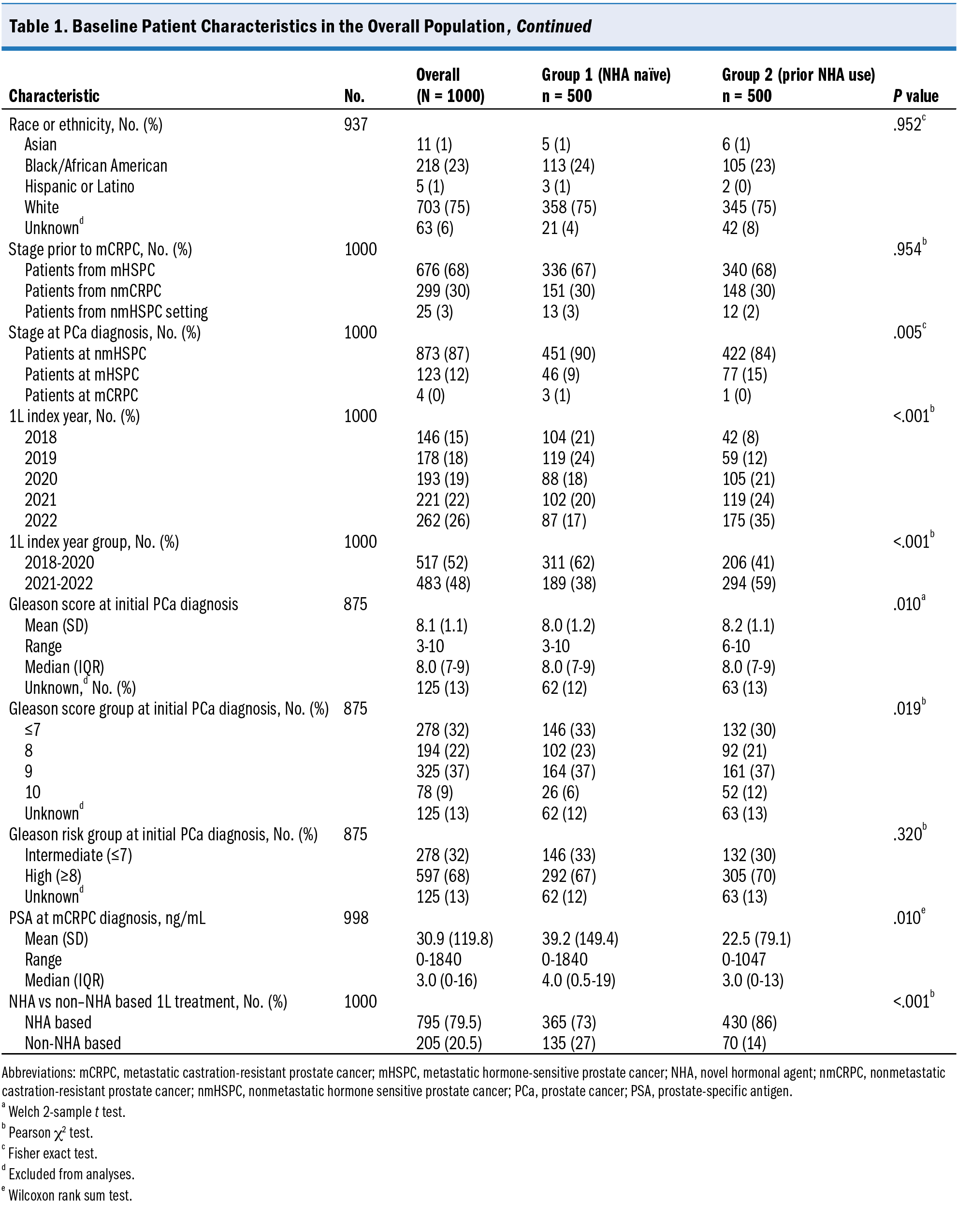
Of the 3300 patients meeting eligibility criteria, 1000 patients were stratified sampled for inclusion in group 1 (NHA naive; n = 500) and group 2 (prior NHA use; n = 500). Patient demographics and baseline characteristics are presented in Table 1. The mean age of the overall population was 75.8 years at the 1L index date; most (703 patients, [75%]) were White, and about one-fourth (218 patients, [23%]) were Black/African American. Among patients with a known Gleason score at initial PCa diagnosis, 68% had a score >7. Geographic region distribution differed between group 1 and group 2 (P = .021). Patients in group 1 were more likely than those in group 2 to have nonmetastatic HSPC at initial PCa diagnosis (90% vs 84%, P = .005). The mean Gleason score at initial PCa diagnosis was slightly higher in group 2 than in group 1 (8.2 vs 8.0, P = .01). The median PSA level at mCRPC diagnosis was 3.0 ng/mL for the overall population. Group 2 had a lower mean PSA level at mCRPC diagnosis than group 1 (22.5 vs 39.2 ng/mL, respectively; P = .01). More patients received NHA-based 1L treatment (79.5%) compared with non–NHA-based treatment (20.5%) in the overall population. Group 2 had a higher proportion of patients who received NHA-based 1L treatment than group 1 (86% vs 73%, respectively; P < .001).
Among this stratified cohort, 444 patients (44%) had genetic testing performed and documented in the chart at any point during the study period. Of these, next-generation sequencing (NGS) was the most common method used, occurring in 90% (n = 398) who had genetic testing documented. Among those who were tested for BRCA with NGS (n = 364), 12% were positive. Among patients who received HRR testing (n = 283), 24% were positive for the mutation.
Patients in this cohort (N = 1000) were treated by 293 unique urology clinicians during the study period. A total of 112 patients (11%) had a documented oncologist-related office visit within 90 days before or after starting 1L treatment for mCRPC, of whom 56 (50%) consulted both a urologist and an oncologist for 1L. An oncologist prescribed 1L for 38 patients.
Social determinants of health (SDOH) information, including household size, education, marital status, and income, was available for only 23% (n = 227) of patients. More than 85% (n = 197) had a household size of 2 or more; 48% (n = 108) had completed college or graduate school, 70% (n = 158) were married, and 69% (n = 157) had an income of $50 000 or more. In terms of baseline characteristics, the mean age was 74.6 years; approximately 50% (n = 113) were from the South treatment region; and the majority had Medicare insurance coverage (n = 183 [82%]) and median (IQR) values for Gleason score and PSA as 8.0 (7-9) and 2.0 (0-13), respectively.
Treatment Patterns
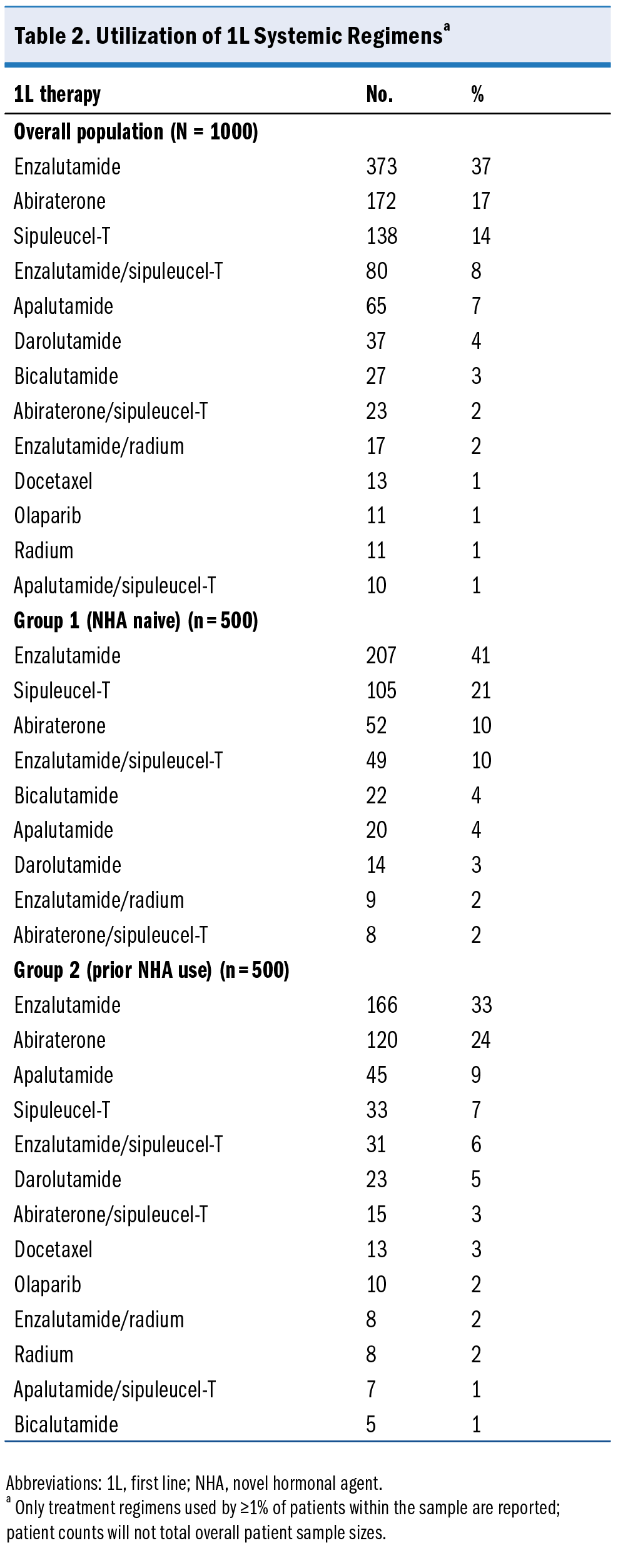
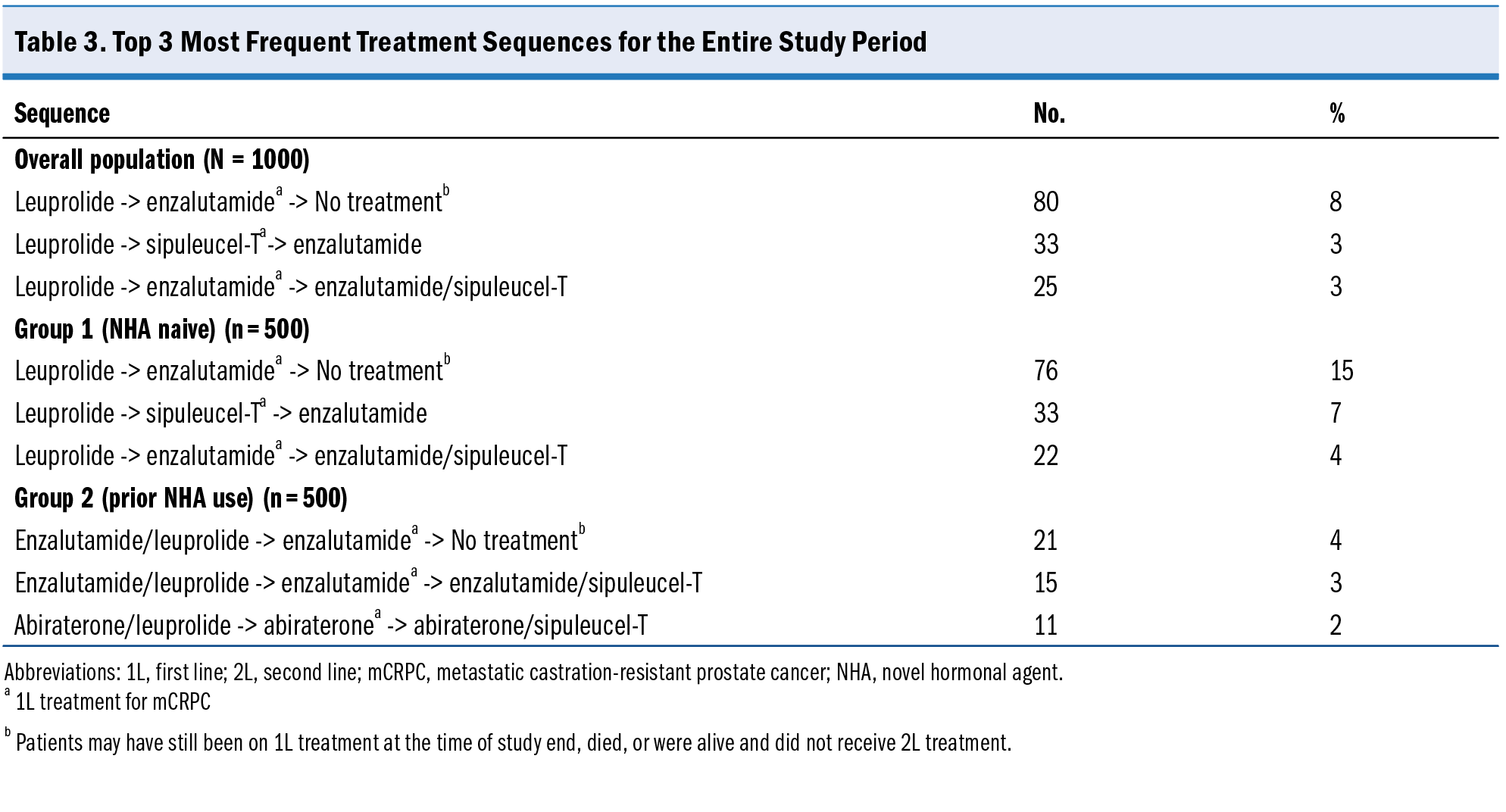
The proportion of patients who were NHA naive at 1L showed a decreasing trend over time: 71% (2018), 67% (2019), 46% (2020), 46% (2021), and 33% (2022). The most common 1L systemic regimens in the overall population (Table 2) were enzalutamide (37%), abiraterone (17%), and sipuleucel-T (14%). Combined, the top 3 1L treatment regimens accounted for 68% of overall 1L therapy, and NHA monotherapy accounted for 65% of 1L therapy. A total of 13 patients in our population received a PARP inhibitor in 1L treatment (11 monotherapy, 2 in combination with an NHA). Refer to Table 3 for the most common treatment sequences. In the overall population, 549 (55%) patients received 2L therapy, 267 (49%) of whom were NHA naive, and 282 (51%) had prior NHA use.
Clinical Outcomes
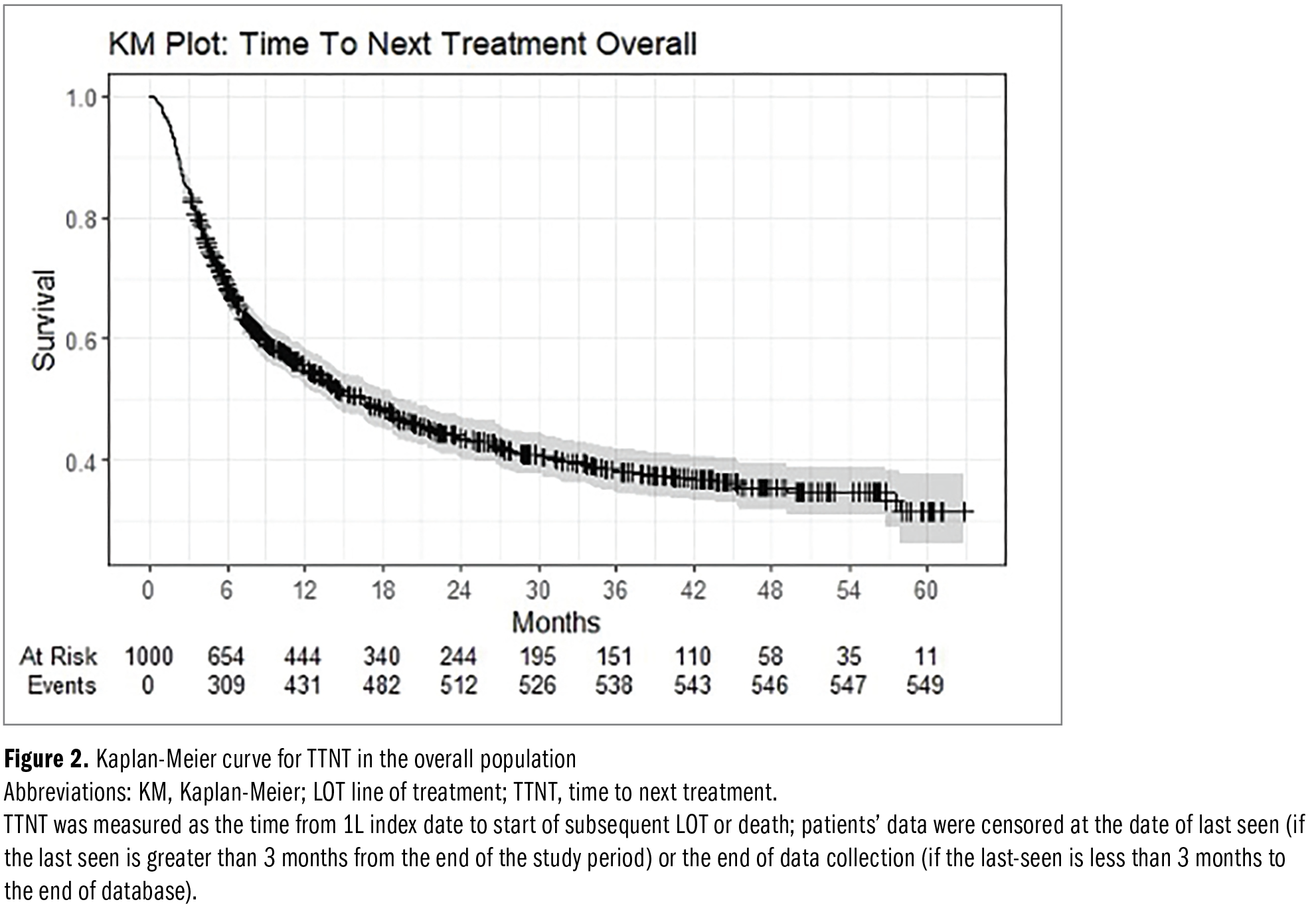
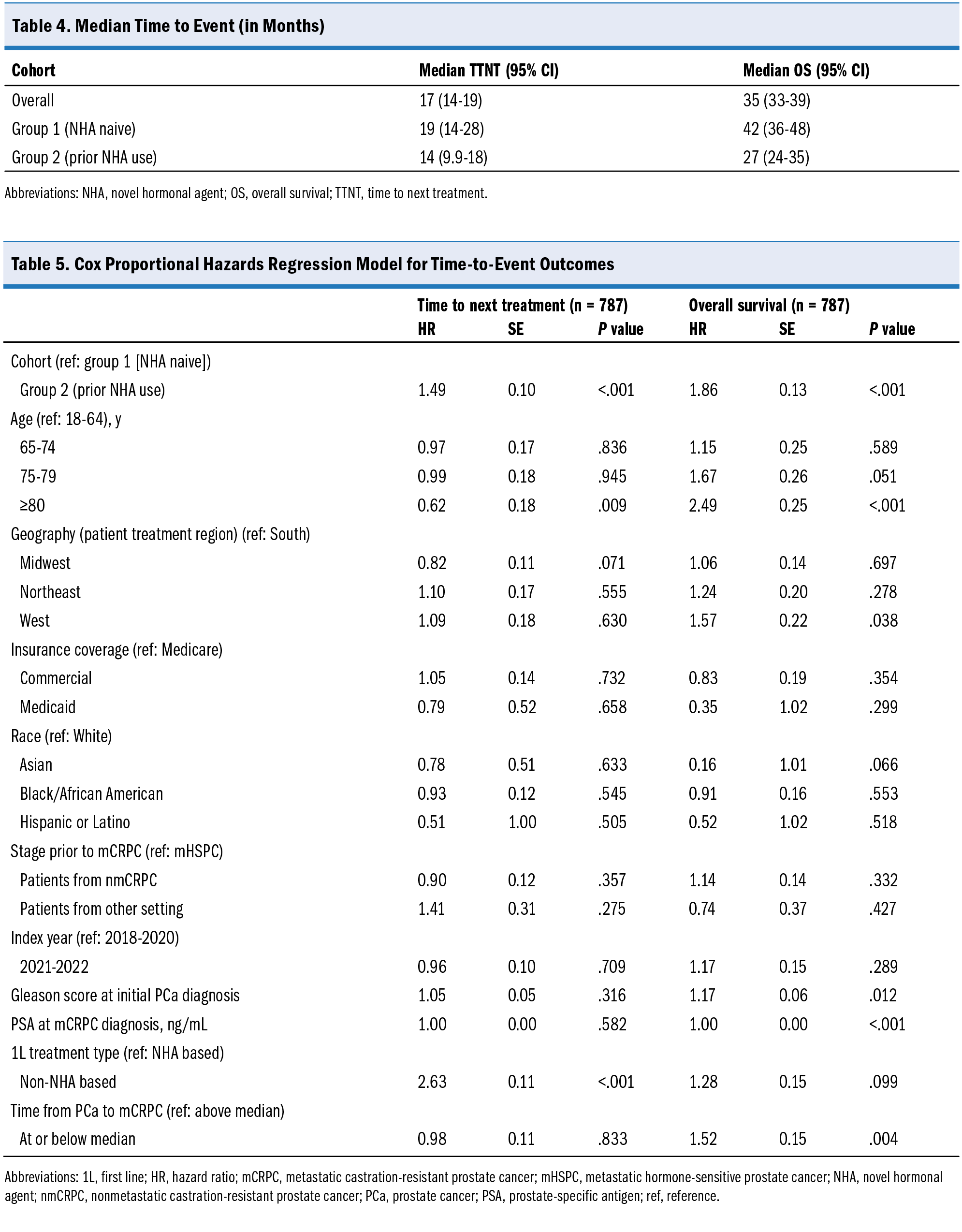
Median TTNT was 17 months in the overall population (95% CI, 14-19 months) (Figure 2). The median TTNT for group 1 was 19 months (95% CI, 14-28 months) and for group 2 was 14 months (95% CI, 10-18 months) (Table 4). Adjusting for clinical and demographic characteristics (Table 5), patients in group 2 were more likely to initiate a new anticancer therapy than patients in group 1 (hazard ratio [HR], 1.49;
P < .001), and patients receiving non–NHA-based 1L treatment were more likely than those receiving NHA-based 1L treatment to initiate new anticancer therapy (HR, 2.63; P < .001). Patients who were 80 years of age or older were less likely to initiate new anticancer therapy than those aged 18 to 64 years (HR, 0.62; P = .009).
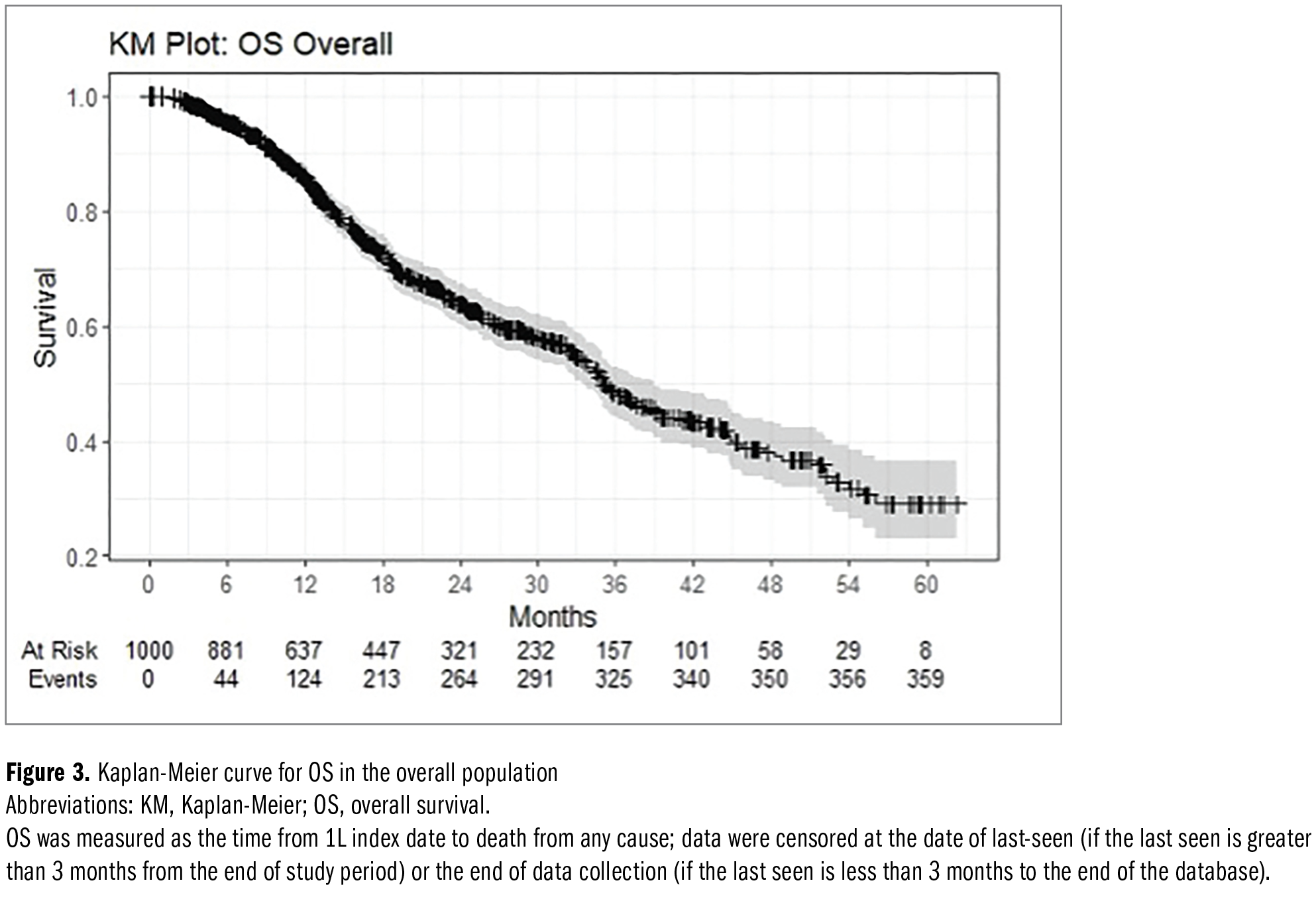
Median OS was 35 months for the overall population (95% CI, 33-39 months) (Figure 3). The median OS for group 1 was 42 months (95% CI, 36-48 months) and for group 2 was 27 months (95% CI, 24-35 months) (Table 4). Adjusting for clinical and demographic characteristics (Table 5), patients in group 2 had a higher risk of death than those in group 1 (HR, 1.86; P < .001). Patients aged 80 years and older had a higher risk of death than those aged 18 to 64 years (HR, 2.49; P < .001), and patients with a time from initial PCa diagnosis to mCRPC at or below median time had a higher risk of death than those with a time from PCa diagnosis to mCRPC that was above the median (HR, 1.52; P = .004). Patients who received non–NHA-based 1L treatment had a numerically higher risk of death than patients who received NHA-based 1L treatment (not statistically significant).
NHA (pre-1L mCRPC) to NHA (1L mCRPC) Analysis
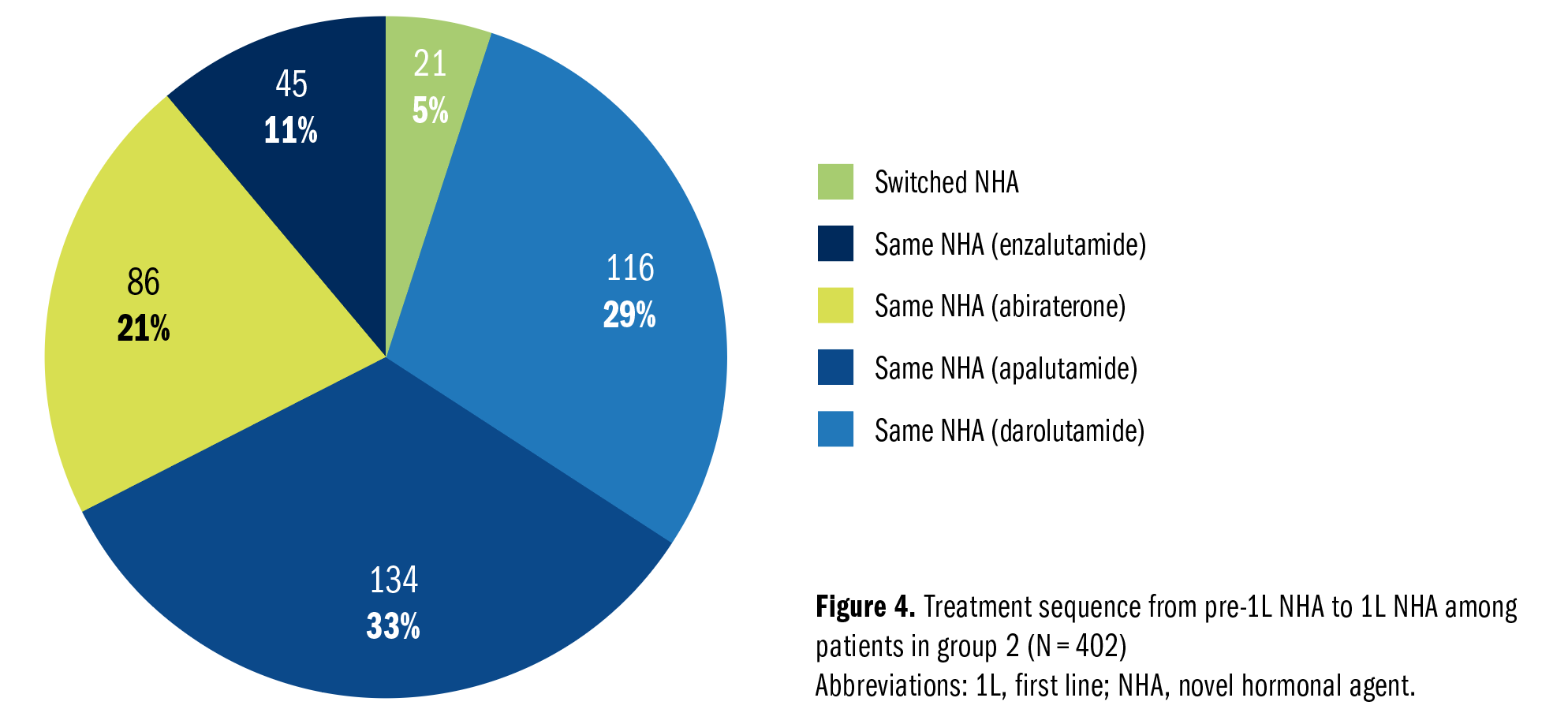
For the 795 patients who were treated with NHA-based treatment in 1L mCRPC, 285 (36%) received NHA for mHSPC. Among patients in group 2 specifically, 402 patients (80%) were treated with an NHA prior to mCRPC and for 1L mCRPC. The treatment sequences for this subgroup are shown in Figure 4, with enzalutamide -> enzalutamide as the most common (33%), followed by abiraterone -> abiraterone (21%), then apalutamide -> apalutamide (11%). Most patients receiving an NHA in both pre-1L and 1L treatment received the same drug (71%) compared with those who switched to a different NHA (29%).
Black/African American Subgroup Analysis
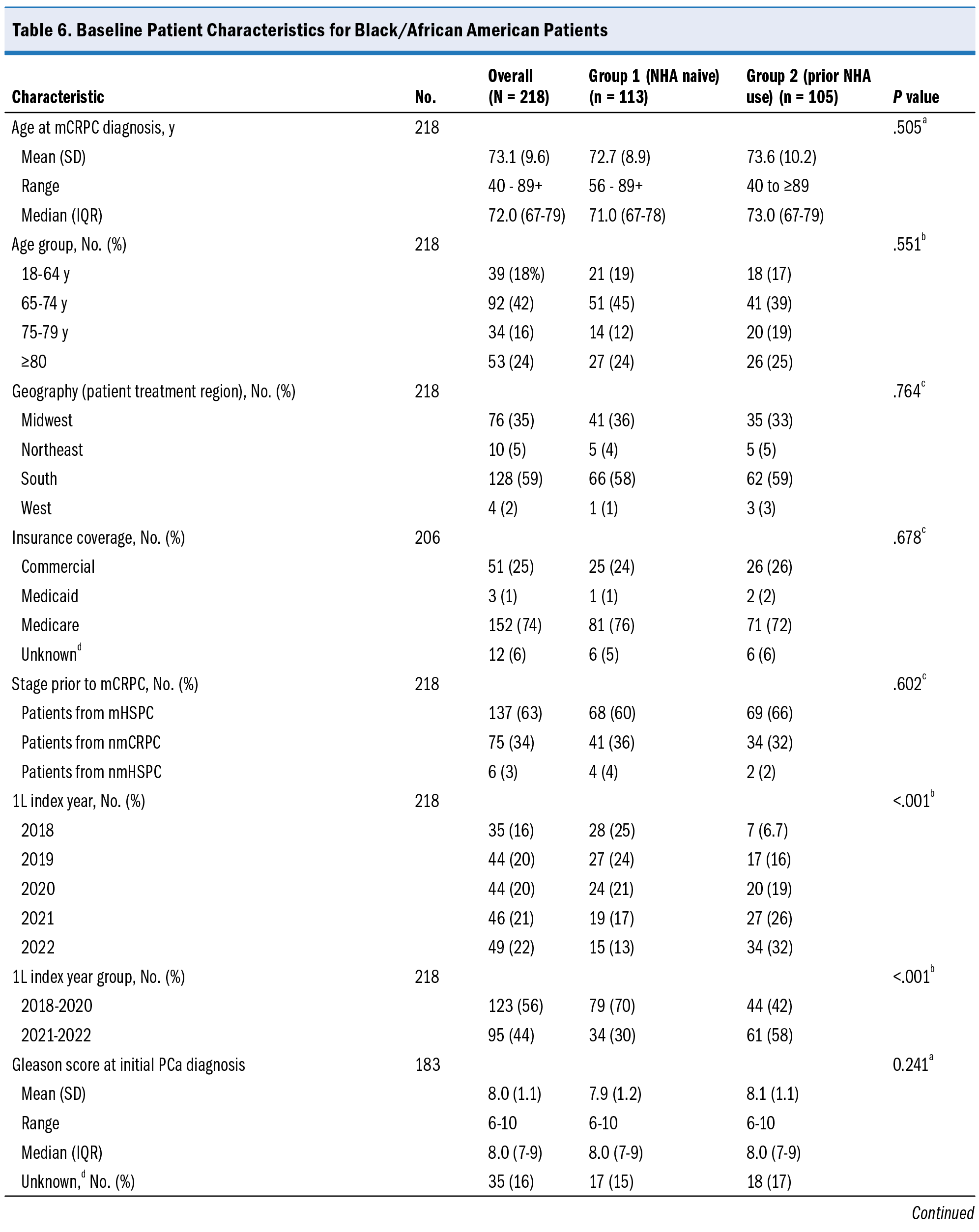
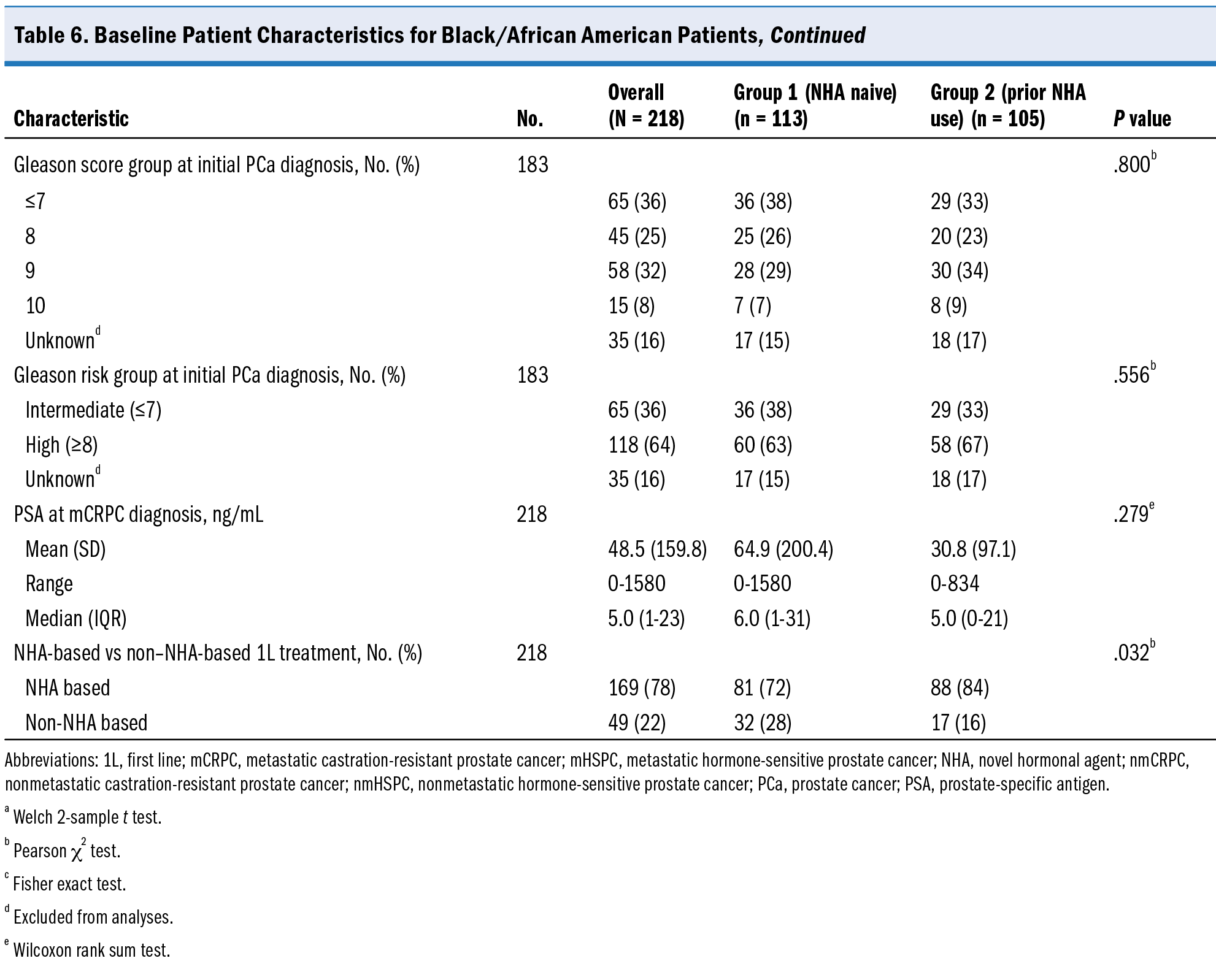
Baseline and demographic characteristics for the Black/African American subcohort (n = 218 [23%]) are presented in Table 6. The mean age was 73.1 years, with 65% having a Gleason score >7 at PCa diagnosis. The median PSA level at mCRPC diagnosis was 5.0 ng/mL, and more patients received NHA-based 1L treatment (78%) than non–NHA-based 1L treatment (22%). Median TTNT from the start of 1L treatment for mCRPC was 27 months (95% CI, 14-57 months), and median OS was 39 months (95% CI, 34 months–not reached).
Discussion
In this real-world, noninterventional analysis, we explored descriptive characteristics, treatment patterns, and clinical outcomes for 1000 patients from US community urology practices who initiated 1L mCRPC therapy between January 1, 2018, and December 31, 2022. The start of this time period coincided approximately with the US Food and Drug Administration (FDA) approval of abiraterone in mHSPC, the first NHA to be approved for patients in this setting; it was anticipated that this approval may begin to shift systemic treatment patterns for mCRPC. In this stratified cohort of patients, most (80%) received NHA-based 1L treatment for mCRPC, and a notable proportion of these patients also received NHA-based treatment in a prior LOT, despite a lack of robust data supporting sequential use of these agents. A key finding from our analysis is that nearly 40% of the overall population were treated with an NHA in both 1L mCRPC and mHSPC. Other real-world studies have reported similar findings, despite a lack of data and guideline recommendations to support this approach, perhaps reflecting the desire by patients and providers to avoid side effects of alternate therapies, including chemotherapy, as well as patient performance status or comorbidities that help inform treatment selection.15-17
Median OS was 35 months for the overall population, and TTNT was 17 months; patients in our study experienced numerically longer OS and TTNT than other real-world studies in mCRPC, likely due to a variety of differing factors, including the severity and aggressiveness of disease related to the cohorts.15-19 The OS in other real-world studies of patients with mCRPC ranged from 12.9 months to 23.7 months, and TTNT was 13.7 months.15-19 These studies included patients identified from the Flatiron Health database and Medicare claims data, respectively. With our population being treated primarily in a urology setting, patients likely experienced better outcomes due to a lower severity of disease compared with patients treated in oncology settings. More effective therapies for mCRPC available in recent years (ie, NHAs) may also have contributed.15-19
Disparities in care across race and ethnicity exist across several different types of cancer; however, Black/African American patients with PCa tend to be differentially impacted given poor access to care and late diagnoses of disease.20,21 A higher percentage of patients in our cohort were Black/African American compared with other observational studies and clinical trials.22,23 Black/African American patients in our study tended to be younger, with more advanced disease at initial diagnosis; however, with treatment, they experienced longer numerical median OS and TTNT relative to the overall population. Similar trends have been observed in other published research. The prospective PANTHER study evaluated outcomes in Black and White men with mCRPC receiving abiraterone, with primary findings showing similar progression-free survival (PFS) and OS rates; however, Black patients achieved higher rates of PSA decline by ≥50% (26% vs 10%) and longer time to PSA progression (16.6 months vs 11.5 months).24 A real-world study analyzing data from Flatiron Health found slightly longer numerical outcomes related to time to treatment discontinuation and OS in Black patients vs the overall population; Black race was also significantly associated with longer time to treatment discontinuation vs patients who were White.16
National Comprehensive Cancer Network guidelines recommend multigene tumor testing for alternations in HRR genes and microsatellite instability-high or deficient mismatch repair in patients with metastatic PCa and mCRPC, respectively.14 Tumor mutational burden testing may be considered in patients with mCRPC. Nearly half of patients’ tumors in our study were tested at any point during the study period utilizing NGS, BRCA, and/or HRR testing platforms. Overall, previously published literature reported underutilization of these tests relative to other types of cancer, including mCRPC and mHSPC.25-27 In analyses of mCRPC data through 2021 from Flatiron Health, barriers to HRR testing included age 65 years or older and treatment within a community oncology setting. Black patients with metastatic PCa were also disproportionately impacted, reinforcing the need for mitigating strategies that facilitate better access to care for these patients. Additionally, Barata et al27 demonstrated that in US patients with mHSPC between January 2020 and August 2020, 72% did not receive HRR mutation testing, and more patients receiving care by an oncologist were tested vs those receiving care by a urologist. A lack of family history of PCa as well as lack of visceral metastases were also associated with a lower likelihood of HRR mutation testing. It is important to understand how testing patterns will continue to evolve over time with not only the availability of current therapies but also the increased need for clinical trial participation and the subsequent, future availability of new therapeutic modalities.
Our population had a relatively low likelihood of receiving multidisciplinary care, which is a consistent pattern with what has been reported in other real-world research in PCa.28-20 Only 11% of patients in our study had a documented oncologist-related office visit. The remainder of the patients were solely cared for by their urologist. The lack of referrals to oncology represents an opportunity to optimize the treatment of patients with mCRPC by engaging a multidisciplinary team.
Several limitations should be considered while interpreting the results of this study. First, data for this 1000-patient cohort may not be generalizable to the broader population of US patients with mCRPC as groups 1 and 2 were created based on specific inclusion and exclusion criteria. Second, data within EMRs were primarily collected for clinical and administrative purposes rather than research; however, we employed standardization measures to ensure consistent use and interpretation of structured and unstructured data and noted missing data, where applicable. Third, measurement of real-world PFS was limited by lack of documentation of progression in the EMR; therefore, TTNT was used as a proxy based on experience in other real-world studies.30-32 Social determinants of health information was available only for 23% of the patients, and further analysis should be strongly considered for future exploration with larger cohorts.
Conclusions
In a population of patients treated by community urologists, a notable percentage of patients received NHA-based treatment for both mHSPC and 1L mCRPC. A small percentage of patients were referred to an oncologist for care. Coinciding with the timing of FDA approval of the first NHA for mHSPC, the percentage of patients who were NHA naive at 1L mCRPC between 2018 and 2022 trended downward with each subsequent year. Among the entire cohort, the top 3 1L treatments for mCRPC were enzalutamide, abiraterone, and sipuleucel-T. Patients receiving NHA-based 1L treatment were less likely to initiate new anticancer therapy during the study period. Black patients experienced numerically better TTNT and OS than the overall population, despite a greater percentage of patients with advanced disease at diagnosis.
Based on these findings, opportunities for improvement in the care of patients with mCRPC within a US community urology network include better adherence to guideline recommendations with respect to use of NHAs, consideration of genetic testing, greater focus of screening efforts for Black patients, and greater multidisciplinary collaboration with oncology clinicians.
References
1. Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209-249. doi:10.3322/caac.21660
2. Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74(1):12-49. doi:10.3322/caac.21820
3. National Cancer Institute. Cancer stat facts: prostate cancer. SEER. Accessed May 14, 2024. https://seer.cancer.gov/statfacts/html/prost.html
4. Centers for Disease Control and Prevention. US cancer statistics prostate cancer stat bite. Updated June 13, 2024. Accessed February 24, 2025. https://www.cdc.gov/united-states-cancer-statistics/publications/prostate-cancer-stat-bite.html
5. Beer TM, Armstrong AJ, Rathkopf DE, et al; PREVAIL Investigators. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med. 2014;371(5):424-433. doi:10.1056/NEJMoa1405095
6. Beer TM, Armstrong AJ, Rathkopf D, et al. Enzalutamide in men with chemotherapy-naïve metastatic castration-resistant prostate cancer: extended analysis of the phase 3 PREVAIL study. Eur Urol. 2017;71(2):151-154. doi:10.1016/j.eururo.2016.07.032
7. Ryan CJ, Smith MR, De Bono JS, et al. Abiraterone in metastatic prostate cancer without previous chemotherapy. N Engl J Med. 2013;368(2):138-148. doi:10.1056/NEJMoa1209096
8. Ryan CJ, Smith MR, Fizazi K, et al. Abiraterone acetate plus prednisone versus placebo plus prednisone in chemotherapy-naive men with metastatic castration-resistant prostate cancer (COU-AA-302): final overall survival analysis of a randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 2015;16(2):152-160. doi:10.1016/S1470-2045(14)71205-7
9. Taylor AK, Kosoff D, Emamekhoo H, Lang JM, Kyriakopoulos CE. PARP inhibitors in metastatic prostate cancer. Front Oncol. 2023;13:1159557. doi:10.3389/fonc.2023.1159557
10. Caram MEV, Burns J, Kumbier K, et al. Factors influencing treatment of veterans with advanced prostate cancer. Cancer. 2021;127(13):2311-2318. doi:10.1002/cncr.33485
11. Malangone-Monaco E, Li W, Noxon V, et al. Real-world treatment patterns among patients with metastatic castration-resistant prostate cancer (mCRPC) in the US. J Clin Oncol. 2022;40(6_suppl):49-49. doi:10.1200/JCO.2022.40.6_suppl.049
12. Barata PC, Leith A, Ribbands A, et al. Treatment patterns among men with metastatic castration-resistant prostate cancer (mCRPC) in the United States (US). J Clin Oncol. 2022;40(6_suppl):52-52. doi:10.1200/JCO.2022.40.6_suppl.052
13. Specialty Networks. Data on file.
14. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Prostate Cancer. Version 3.2024. March 8, 2024. Accessed May 14, 2024. https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf
15. Shore ND, Laliberté F, Ionescu-Ittu R, et al. Real-world treatment patterns and overall survival of patients with metastatic castration-resistant prostate cancer in the US prior to PARP inhibitors. Adv Ther. 2021;38(8):4520-4540. doi:10.1007/s12325-021-01823-6
16. Smyth EN, Whipple S, Guinter M, et al. Real-world treatment (Tx) sequences and time to discontinuation (rwTTD) in the first-line (1L) metastatic castration-resistant prostate cancer (mCRPC) setting. J Clin Oncol. 2024;42(4_suppl):55-55. doi:10.1200/JCO.2024.42.4_suppl.55
17. George DJ, Sartor O, Miller K, et al. Treatment patterns and outcomes in patients with metastatic castration-resistant prostate cancer in a real-world clinical practice setting in the United States. Clin Genitourin Cancer. 2020;18(4):284-294. doi:10.1016/j.clgc.2019.12.019
18. Freedland SJ, Davis M, Epstein AJ, Arondekar B, Ivanova JI. Real-world treatment patterns and overall survival among men with metastatic castration-resistant prostate cancer (mCRPC) in the US Medicare population. Prostate Cancer Prostatic Dis. 2024;27(2):327-333. doi:10.1038/s41391-023-00725-8
19. George DJ, Agarwal N, Sartor O, et al. Real-world patient characteristics associated with survival of 2 years or more after radium-223 treatment for metastatic castration-resistant prostate cancer (EPIX study). Prostate Cancer Prostatic Dis. 2022;25(2):306-313. doi:10.1038/s41391-021-00488-0
20. Zavala VA, Bracci PM, Carethers JM, et al. Cancer health disparities in racial/ethnic minorities in the United States. Br J Cancer. 2021;124(2):315-332. doi:10.1038/s41416-020-01038-6
21. Chowdhury-Paulino IM, Ericsson C, Vince R, Spratt DE, George DJ, Mucci LA. Racial disparities in prostate cancer among Black men: epidemiology and outcomes. Prostate Cancer Prostatic Dis. 2022;25(3):397-402. doi:10.1038/s41391-021-00451-z
22. Wang WJ, Ramsey SD, Bennette CS, Bansal A. Racial disparities in access to prostate cancer clinical trials: a county-level analysis. JNCI Cancer Spectr. 2022;6(1):pkab093. doi:10.1093/jncics/pkab093
23. Lillard JW, Moses KA, Mahal BA, George DJ. Racial disparities in Black men with prostate cancer: a literature review. Cancer. 2022;128(21):3787-3795. doi:10.1002/cncr.34433
24. George DJ, Halabi S, Heath EI, et al. A prospective trial of abiraterone acetate plus prednisone in Black and White men with metastatic castrate-resistant prostate cancer. Cancer. 2021;127(16):2954-2965. doi:10.1002/cncr.33589
25. Ikeda S, Elkin SK, Tomson BN, Carter JL, Kurzrock R. Next-generation sequencing of prostate cancer: genomic and pathway alterations, potential actionability patterns, and relative rate of use of clinical-grade testing. Cancer Biol Ther. 2019;20(2):219-226. doi:10.1080/15384047.2018.1523849
26. Barata PC, Assayag J, Li B, Siu G, Nazari J, Niyazov A. Real-world homologous recombination repair gene mutation testing trends in patients with metastatic castration-resistant prostate cancer in the United States. Poster presented at: American Society of Clinical Oncology Genitourinary Cancers Symposium; February 16-28, 2023; San Francisco, CA. Poster 98.
27. Barata P, Montgomery R, Gillespie-Akar L, Nazari J, Arondekar B, Niyazov A. Real-world homologous recombination repair mutation testing patterns in patients with metastatic castration-sensitive prostate cancer in the US. Poster presented at: American Society of Clinical Oncology Genitourinary Cancers Symposium; February 16-28, 2023; San Francisco, CA. Poster 99.
28. Gajra A, Hime S, Jeune-Smith J, Russell A, Feinberg B. Referral patterns and treatment preferences in patients with advanced prostate cancer (aPC): differences between medical oncologists and urologists. J Clin Oncol. 2021;39(6_suppl):71-71. doi:10.1200/JCO.2021.39.6_suppl.71
29. Yong C, Onukwugha E, Mullins D, et al. Physician referral patterns among men with advanced prostate cancer. J Clin Oncol. 2014;32(4_suppl):211-211. doi:10.1200/jco.2014.32.4_suppl.211
30. Walker MS, Herms L, Miller PJE. Performance of time to discontinuation and time to next treatment as proxy measures compared with direct observation of progression-free survival. Poster presented at: International Society for Pharmacoeconomics and Outcomes Research Annual Meeting [virtual]; May 18-20, 2020. Poster 284.
31. Walker B, Boyd M, Aguilar K, et al. Comparisons of real-world time-to-event end points in oncology research. JCO Clin Cancer Inform. 2021;5:45-46. doi:10.1200/CCI.20.00125
32. Campbell BA, Scarisbrick JJ, Kim YH, Wilcox RA, McCormack C, Prince HM. Time to next treatment as a meaningful endpoint for trials of primary cutaneous lymphoma. Cancers (Basel). 2020;12(8):2311. doi:10.3390/cancers12082311
Article Information
Published: 3/31/2025.
Conflict of Interest Disclosures: E. N. Smyth, S. Whipple, Z. L. Cui, N. Haddad, and A. V. Balar are employed by and own stock in Eli Lilly and Company. N. Lazarou, K. Grant, and A. Kashyap are employed by Specialty Networks, a Cardinal Health company. N. D. Shore has received fees from Amgen, AstraZeneca, Bayer, Dendreon, Ferring Pharmaceuticals, Genentech, Janssen Scientific Affairs, Medivation/Astellas, Myovant Sciences, Pfizer, and Tolmar. Specialty Networks and PPS Analytics were responsible for the study design in collaboration with Eli Lilly and Company; data management and statistical analysis were led by Specialty Networks and PPS Analytics. Data interpretation and development of this manuscript were completed in collaboration with all authors.
Funding/Support: This study was sponsored by Eli Lilly and Company. The funder supported development of the manuscript through provision of medical writing assistance.
Author Contributions: All authors had the final responsibility for the decision to submit for publication.
Data Availability Statement: The data on file presented in this article are proprietary and not publicly available.
Acknowledgments: The authors thank Austin Ulrich, PharmD, BCACP, of Dragonfly Editorial for writing assistance.
Supplementary Material: Supplementary material is available at Reviews in Urology online.