Introduction
Prostate cancer is the most prevalent noncutaneous malignancy in the world and a leading cause of cancer-related deaths in men.1 Substantial increases in the rates of prostate cancer diagnosis and cancer-related deaths over the next 2 decades are predicted.2 The majority of men diagnosed with prostate cancer present with either nonmetastatic or localized disease.3 The importance of appropriate risk stratification cannot be overstated because risk stratification informs the need for further diagnostics and treatments as well as the risk of progression or recurrence.4
Key Points
- Men with very high-risk prostate cancer in our cohort had low rates of receiving treatment intensification with ABI.
- Patients who had 2 or more very high-risk features were more likely to receive treatment intensification with ABI, suggesting that perhaps patients with only 1 qualifying very high-risk feature were not appropriately risk stratified in our study.
- Patients diagnosed in 2023 or 2024 were more likely to receive appropriate treatment intensification with ABI, perhaps reflecting the delay in clinical application from publication of clinical trials.
Abbreviations
ABI abiraterone acetate
NCCN National Comprehensive Cancer Network
RT radiation therapy
STAMPEDE Systemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation or Drug Efficacy
Most men who die from prostate cancer are diagnosed with nonmetastatic disease at the outset.5 Among men with high-risk or very high-risk localized prostate cancer, however, there is a clinically significant likelihood of metastasis or recurrence. Roughly 20% to 30% of prostate cancer diagnoses fall into the high-risk category at the time of biopsy.6 Within this subset, some men present with very high-risk disease, characterized either by 2 or 3 high-risk features (prostate-specific antigen value >20 ng/mL, Grade Group 4 disease, T3a) or by 1 or more very high-risk features (>4 cores scored at Grade Group 4, T3b disease, primary Gleason 5 disease).4 For men diagnosed with aggressive localized prostate cancer, delaying or preventing metastasis is paramount because of its association with a worse prognosis.7,8
In the case of men with very high-risk disease, studies have shown that in men opting for radiation therapy (RT), treatment intensification with the addition of abiraterone acetate (ABI) 1000 mg daily and prednisolone 5 mg daily improves metastasis-free and overall survival.9 Identifying men with very high-risk disease can be difficult, however, because it necessitates the integration of structured and unstructured data from large electronic health record datasets. Consequently, there is a risk that men at the highest risk of metastasis and metastasis-related events may not be offered treatment intensification.
Our study delved into the use of treatment intensification with ABI in men with very high-risk, clinically localized prostate cancer who opted for RT within a large urology group practice.
Methods
Patients newly diagnosed with prostate cancer between January 2022 and January 2024 were identified through a retrospective review of a large urology group practice’s clinical navigation pathway. Nurse navigators manually converted all data from an unstructured into a semistructured format. Data were input into a navigation program internally developed to identify patients with high-risk or very high-risk prostate cancer. Each chart of a patient with high-risk or very high-risk prostate cancer underwent manual review to ensure accuracy. Men were categorized as having very high-risk, clinically localized prostate cancer if they were older than 18 years of age and had 2 or more high-risk features (prostate-specific antigen level >20 ng/mL, Grade Group 4 disease, T3a) or any very high-risk feature (>4 cores of Grade Group 4 or disease, T3b disease, or primary Gleason score 5 disease). Pathology reports and magnetic resonance imaging scans were manually reviewed to convert unstructured data into structured data. Extracapsular extension and seminal vesicle invasion were considered present only when explicitly stated by the radiologist; terms such as capsular bulging or capsular abutment were not considered indicative of extracapsular disease.
Patients with metastatic disease identified through conventional or prostate-specific membrane antigen imaging as well as individuals who left the practice before treatment initiation were excluded from our analysis. Patient demographic and medical comorbidity data, treatment plans, and urologist characteristics were collected. The analytic cohort included only patients who underwent RT with androgen-deprivation therapy administration.
Descriptive statistics were used to summarize the data, with discrete values presented as percentages or frequencies and continuous variables described by mean and SD. The statistical significance between categorical variables was determined using the χ2 test, and the statistical significance between continuous variables was assessed using the t test. This study was approved by our institutional review board (No. E-23-5351).
Results
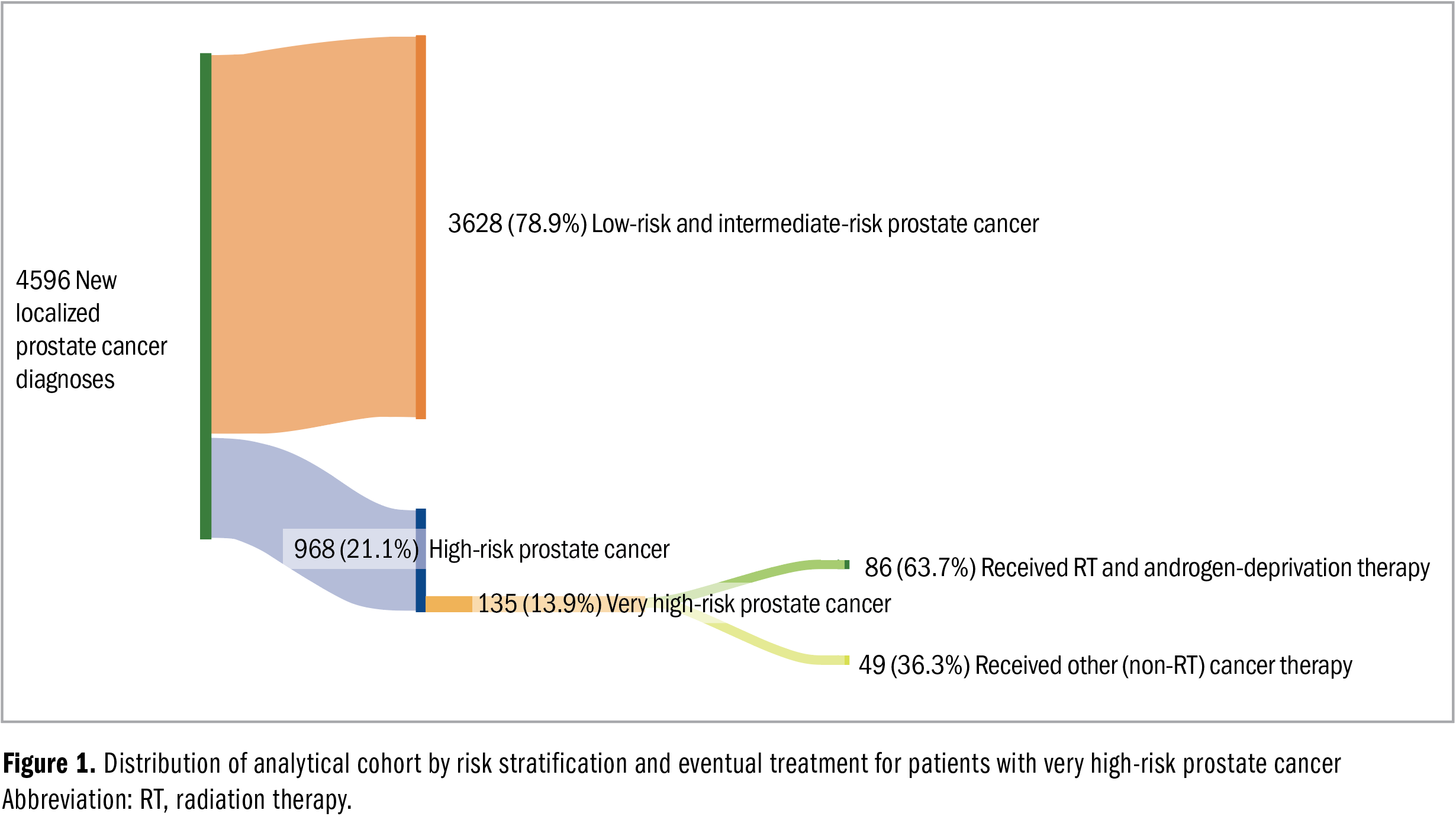
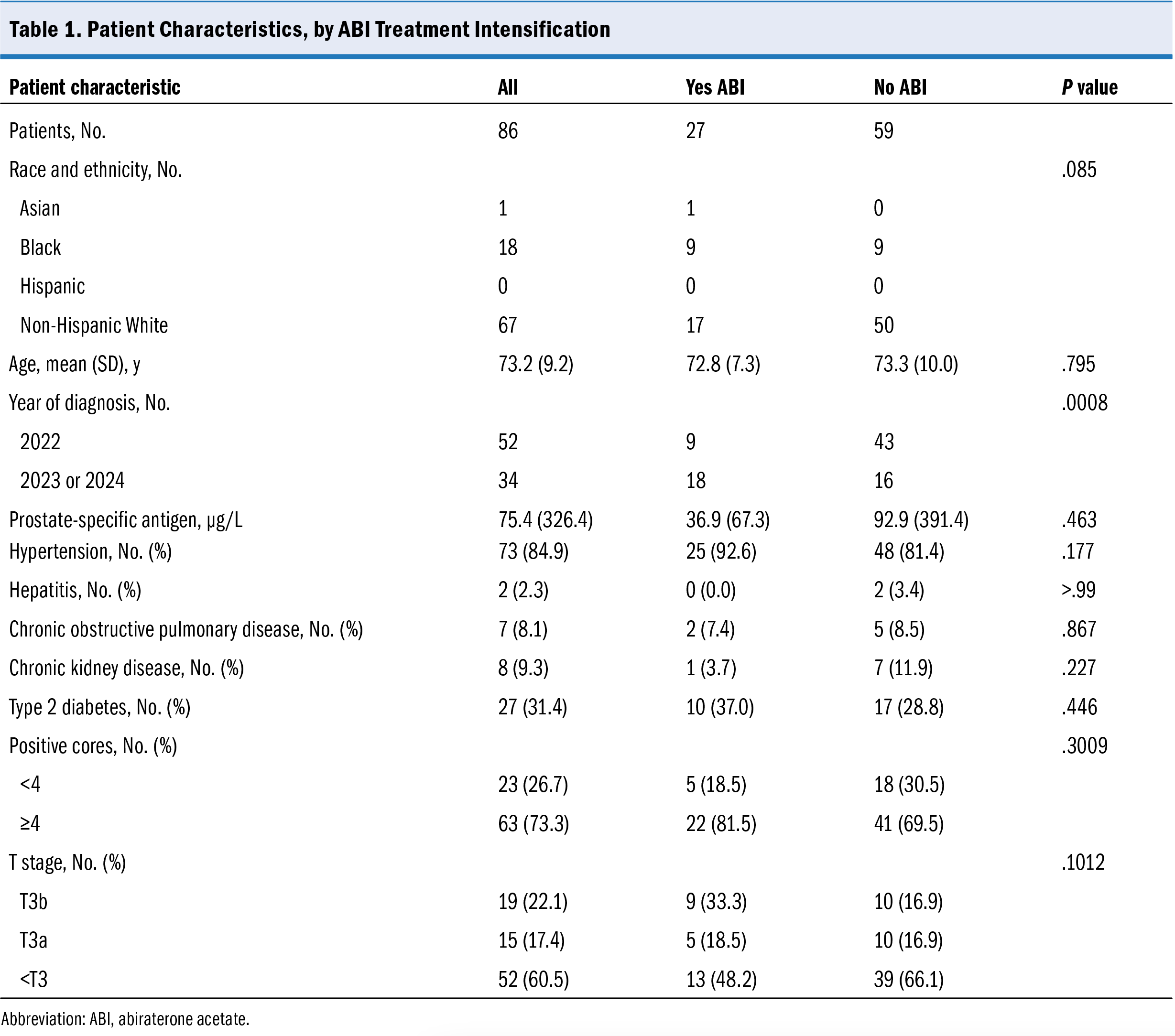
During the study period, 4596 patients were identified as having a positive biopsy. Among them, 968 (21.1%) were diagnosed with localized, high-risk prostate cancer without evidence of extraprostatic disease on metastatic evaluation. Further categorization of the patients with high-risk prostate cancer revealed 135 (13.9%) men with very high-risk disease, with 86 (63.7%) of them undergoing treatment with RT and androgen-deprivation therapy as the primary treatment modality, as illustrated in Figure 1. The characteristics of the cohort, including oncologic and medical comorbidities, are detailed in Table 1. The mean (SD) age of these patients was 73.2 (9.2) years, and the mean (SD) prostate-specific antigen level was 75.4 (326.4) ng/mL.
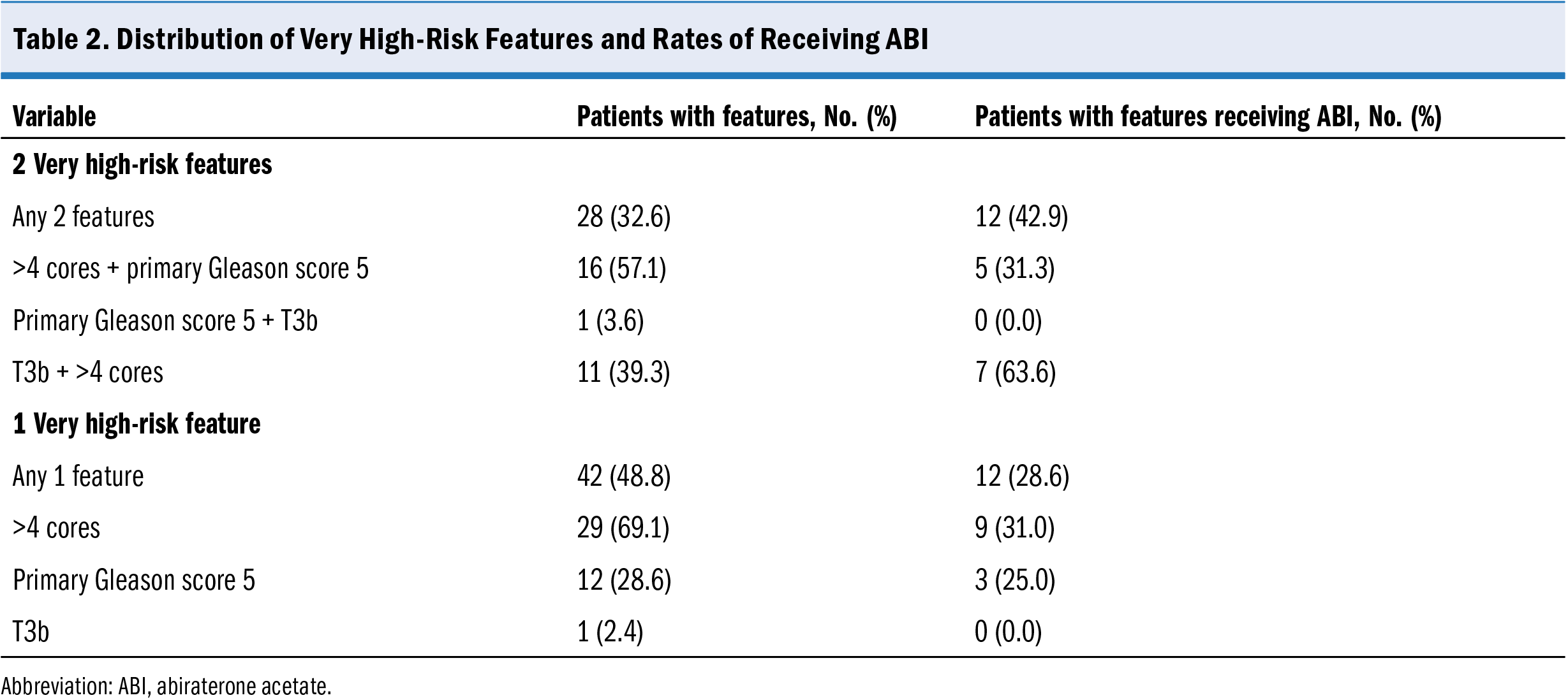
Table 2 lists the distribution and frequency of high-risk and very high-risk features. All patients but 1 presented with Grade Group 4 (33/86 [38.4%]) or Grade Group 5 (52/86 [60.5%]) disease. The majority of the analytic cohort (73.26%) met very high-risk disease criteria by having more than 4 cores positive for Grade Group 4 or higher disease. Another 22.1% met very high-risk disease criteria by being identified with T3b disease during the staging workup. The remaining 4.7% of patients fulfilled very high-risk disease criteria by exhibiting 2 or more high-risk features. Of the 86 eligible patients, 27 (31.4%) received ABI. No statistically significant differences were observed in race or ethnicity, age, medical comorbidities, number of cores positive for Grade Group 4 or higher disease, or T stage at diagnosis between patients who did and did not receive ABI. Being diagnosed in 2023 or 2024 was associated with higher rates of receiving ABI than being
diagnosed in 2022 (P = .0008).
A subgroup analysis was conducted based on the number of very high-risk disease factors present. A total of 42 patients exhibited just 1 very high-risk disease factor, with the number of positive cores being the most prevalent qualifying factor. Among these patients, only 12 (28.6%) received ABI. The rate of ABI administration increased to 42.9% for patients with 2 very high-risk disease features, although this difference was not statistically significant (P = .30).
Twenty-eight patients had 2 very high-risk disease factors, with the combination of more than 4 cores of Grade Group 4 or higher and primary Gleason score 5 disease being the most common (57.1%), followed by stage T3b with more than 4 cores of Grade Group 4 or higher (39.3%). Interestingly, among individuals with more than 4 cores of Grade Group 4 or higher combined with primary Gleason score 5 disease, only 31.3% received ABI. In contrast, among patients with more than 4 cores of Grade Group 4 or higher and T3b staging, 63.6% received ABI. This disparity, however, did not achieve statistical significance (P = .13).
Discussion
In this real-world study, we found that 31.4% of men with very high-risk, clinically localized prostate cancer who chose RT received treatment intensification with ABI and androgen-deprivation therapy. To retrospectively identify very high-risk prostate cancer cases, nurse navigators converted unstructured data into structured data, which were then input into an internally developed program to identify patients with very high-risk prostate cancer. Analysis of patient, urologist, and oncologic factors did not reveal specific attributes associated with the low rates of ABI administration. This finding suggests that men at high risk of recurrence or metastasis are not receiving treatment intensification. Our data indicate that multiple factors affect treatment selection, including difficulty in identifying patients with very high-risk prostate cancer and understanding current clinical guidelines.
The benefit of treatment intensification for men with very high-risk prostate cancer is well established. The Systemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation or Drug Efficacy (STAMPEDE) trial (ClinicalTrials.gov identifier NCT00268476) demonstrated that adding ABI to a treatment regimen for nonmetastatic very high-risk prostate cancer increases metastasis-free and overall survival.10 In addition, a pooled analysis of 10 clinical trials that included 3604 patients demonstrated that men with 2 or 3 high-risk factors and patients with nodal involvement have worse outcomes than men with only 1 high-risk factor. Specifically, metastasis-free survival, time to metastasis, prostate cancer–specific mortality, and overall survival were all worse in men with 2 or 3 high-risk factors (very high-risk, clinically localized disease) or locoregional disease.11 Metastasis-related events, including skeletal-related events, can severely reduce patient quality of life, causing pain, fatigue, and urinary and sexual dysfunction.12 Despite strong evidence supporting treatment intensification, however, our study shows that many men do not receive appropriate treatment.
Classifying a patient as having very high-risk prostate cancer is complex. One must synthesize information from multiple records, including pathology and imaging reports, often dealing with unstructured data. Even with all the collected data, risk stratification can be ambiguous. For instance, National Comprehensive Cancer Network (NCCN) guidelines state that “two or three” high-risk features upgrade a patient to very high-risk prostate cancer, allowing for clinical discretion. This decision-making process leads to varied outcomes and prescribing patterns, as seen in our study.
Other potential reasons for low utilization of treatment intensification include the recent incorporation of ABI into clinical guidelines, lack of physician familiarity with guidelines, and the generic nature of the drug. The NCCN guidelines were updated to include ABI for very high-risk disease in September 2021.13 Results from STAMPEDE were published in late December 2021. Other retrospective studies examining treatment intensification also found low utilization after initial approval, with improvement over time.14 The low utilization observed in our study could be due in part to the proximity of the STAMPEDE publication and incorporation into NCCN guidelines to our data-collection period. Increased utilization was seen over time, supporting low initial familiarity with guideline changes. This increased utilization is consistent with our findings, which demonstrate higher utilization of ABI in 2023 and 2024 than in 2022. Our results may reflect low familiarity with guideline changes compounded by decreased industry support because ABI had become generic at the time of publication of STAMPEDE and incorporation into guidelines.15
Prostate cancer screening, staging, and treatment recommendations are dynamic, focusing on targeting treatment efforts for individuals who would benefit the most. In particular, the introduction of improved staging with prostate-specific membrane antigen positron emission tomography/computed tomography imaging has led to more accurate detection of localized high-risk prostate cancer.16 In our study, 21.1% of patients were high risk, and 13.9% of those patients (2.9% of the total) met very high-risk criteria. Previous research had found that 22.5% of patients were high risk. Some studies had excluded patients with very high-risk disease or did not separate the high-risk category. Despite variability in patient categorization and data presentation, the percentage of patients with high-risk disease is generally consistent across studies.
The consistency of risk classification in our patient population with other studies supports the general applicability of our findings. This congruity may change, however, because prostate cancer incidence is projected to rise through 2040, potentially altering the risk-stratification breakdown. Since 2000, high-risk groups have remained a consistent or increasing portion of the population, with improved treatments extending survival and increasing the prevalence of patients with high-risk disease.17,18
More work is needed to document trends of treatment intensification in the population with very high-risk disease. In addition, our group plans to use the methods from this study to identify future patients with very high-risk disease in real time. Nurse navigators will collect all data points and synthesize the information after every biopsy. Men found to have very high-risk disease will be identified before meeting with their urologist to review biopsy data. We hope this process will allow for a more productive physician-patient interaction and increased use of treatment intensification when appropriate.
The limitations of this study include those inherent in a retrospective cohort study. Data were collected directly from patient records, and certain important pieces of information may have been missing. The decision-making process for not prescribing ABI was not indicated in the documentation, and it is possible that some patients declined the additional treatment, despite recommendations from their urologists. Furthermore, not all urologists may be comfortable prescribing ABI, although it may be prescribed by an outside clinician, a fact that could have been omitted from the electronic health record. Our results describe the findings from the multicenter study cohort and may not be applicable to the wider population.
Conclusions
Our study shows that despite strong clinical trial support and inclusion in treatment guidelines, a majority of patients with very high-risk, clinically localized prostate cancer who elected RT were not offered treatment intensification with ABI. Factors that affect low treatment intensification could include low physician awareness of guideline changes, the proximity of our data-collection period to the inclusion of ABI in NCCN treatment guidelines, and difficulties in identifying this patient group.
References
1. Key statistics for prostate cancer. American Cancer Society. Accessed January 19, 2024. https://www.cancer.org/cancer/types/prostate-cancer/about/key-statistics.html
2. James ND, Tannock I, N’Dow J, et al. The Lancet Commission on prostate cancer: planning for the surge in cases. Lancet. 2024;403(10437):1683-1722. doi:10.1016/S0140-6736(24)00651-2
3. Prostate cancer prognosis. Johns Hopkins Medicine. Accessed November 6, 2023. https://www.hopkinsmedicine.org/health/conditions-and-diseases/prostate-cancer/prostate-cancer-prognosis
4. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Prostate cancer. Version 4.2024. National Comprehensive Cancer Network; 2024.
5. Helgstrand JT, Røder MA, Klemann N, et al. Diagnostic characteristics of lethal prostate cancer. Eur J Cancer. 2017;84:18-26. doi:10.1016/j.ejca.2017.07.007
6. Wenzel M, Würnschimmel C, Ruvolo CC, et al. Increasing rates of NCCN high and very high-risk prostate cancer versus number of prostate biopsy cores. Prostate. 2021;81:874-881. doi:10.1002/pros.24184
7. Gandaglia G, Abdollah F, Schiffmann J, et al. Distribution of metastatic sites in patients with prostate cancer: a population-based analysis. Prostate. 2014;74(2):210-216. doi:10.1002/pros.22742
8. Gandaglia G, Karakiewicz PI, Briganti A, et al. Impact of the site of metastases on survival in patients with metastatic prostate cancer. Eur Urol. 2015;68(2):325-334. doi:10.1016/j.eururo.2014.07.020
9. James ND, de Bono JS, Spears MR, et al. Abiraterone for prostate cancer not previously treated with hormone therapy. N Engl J Med. 2017;377(4):338-351. doi:10.1056/NEJMoa1702900
10. Attard G, Murphy L, Clarke NW, et al. Abiraterone acetate and prednisolone with or without enzalutamide for high-risk non-metastatic prostate cancer: a meta-analysis of primary results from two randomised controlled phase 3 trials of the STAMPEDE platform protocol. Lancet. 2022;399(10323):447-460. doi:10.1016/S0140-6736(21)02437-5
11. Ravi P, Xie W, Buyse M, et al. Refining risk stratification of high-risk and locoregional prostate cancer: a pooled analysis of randomized trials. Eur Urol. 2025;87(2):217-224. doi:10.1016/j.eururo.2024.04.038
12. Holm M, Doveson S, Lindqvist O, et al. Quality of life in men with metastatic prostate cancer in their final years before death: a retrospective analysis of prospective data. BMC Palliat Care. 2018;17:126. doi:10.1186/s12904-018-0381-6
13. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Prostate cancer. Version 1.2022. National Comprehensive Cancer Network; 2021.
14. George DJ, Agarwal N, Ramaswamy K, et al. Emerging racial disparities among Medicare beneficiaries and veterans with metastatic castration-sensitive prostate cancer. Prostate Cancer Prostatic Dis. 2024;27(4):765-775. doi:10.1038/s41391-024-00815-1
15. Indurlal P, Ives H, Garey JS, McGuinness M, Pacheco AV, Wilfong LS. Financial impact of generic therapeutic interchange of abiraterone in the oncology care model for the U.S. Oncology Network. J Clin Oncol. 2022;40(16_suppl). doi:10.1200/JCO.2022.40.16_suppl.e17019
16. Hofman MS, Lawrentschuk N, Francis RJ, et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): a prospective, randomised, multicentre study. Lancet. 2020;395(10231):1208-1216. doi:10.1016/S0140-6736(20)30314-7
17. Rawla P. Epidemiology of prostate cancer. World J Oncol. 2019;10(2):63-89. doi:10.14740/wjon1191
18. Cooperberg MR, Cowan J, Broering JM, Carroll PR. High-risk prostate cancer in the United States, 1990-2007. World J Urol. 2008;26(3):211-218. doi:10.1007/s00345-008-0250-7
Article Information
Published: 3/31/2025.
Conflict of Interest Disclosures: Dr Gregory McMahon has been a paid consultant for the following companies within the past 12 months: Johnson & Johnson, Bayer, and Astellas.
Funding/Support: No funding was received for this study.
Author Contributions: All authors were involved in study conception and design, data analysis and interpretation, drafting the manuscript, and critically revising the draft for intellectual content. All authors approved the final version of the article to be published and agree to be accountable for all aspects of the work.
Data Availability Statement: Deidentified data can be made available by the authors upon reasonable request.