Introduction
Prostate cancer remains a leading cause of cancer-related mortality among men worldwide, having caused an estimated 385 000 deaths worldwide, based on GLOBOCAN 2018 data.1 With the population in the United States aging, the rates of new prostate cancer diagnoses and mortality are expected to increase over the next few decades.2 Although localized prostate cancer is often curable, advanced-staged disease pose a substantial clinical challenge.3 Throughout the prostate cancer landscape there has been a striking increase in the number of guideline-endorsed indications for radiation therapy (RT). This increase includes definitive and postoperative management with curative intent; RT to the primary tumor in metastatic hormone-sensitive prostate cancer (mHSPC); metastasis-directed RT in mHSPC and metastatic castration-resistant prostate cancer (mCRPC); and the expanding role of radiopharmaceutical therapy, such as radium-223 dichloride (Ra-223) and lutetium-177 vipivotide tetraxetan prostate-specific membrane antigen therapy (177Lu-PSMA).4-10
The progression to mCRPC marks a critical juncture in treatment as the disease becomes resistant to androgen-deprivation therapy (ADT).11,12 Bone metastases are a hallmark of advanced prostate cancer, affecting more than 90% of patients with mCRPC.13 These metastases contribute substantially to morbidity and mortality, manifesting as skeletal-related events such as pathological fractures, spinal cord compression, and intractable bone pain. Addressing bone health and controlling metastatic disease are key therapeutic goals in this population. Analogous to evolving indications of RT from palliation to improving survival over time, radiopharmaceutical therapy indications have also witnessed substantial expansion. Historical radiopharmaceutical therapies such as strontium and samarium were used for symptomatic palliation.
Radium-223 become the first radiopharmaceutical therapy in the treatment of mCRPC that not only provided symptomatic relief but also improved overall survival and was added to the armamentarium of life-prolonging therapies.5,14-16
The introduction of Ra-223, an α-particle–emitting radioisotope, ushered in a new era in the therapeutic landscape for patients with mCRPC, especially in patients with bone-predominant disease. Approved by the US Food and Drug Administration in 2013, Ra-223 has been incorporated into clinical practice based on the pivotal ALSYMPCA trial, which demonstrated survival benefits and symptom palliation.5 At present, the landscape of treatment options in mCRPC has become substantially complicated compared with the handful of options available historically. These options include multiple life-prolonging therapies, such as androgen receptor pathway inhibitors (ARPIs), taxane-based chemotherapy, immunotherapy (ie, sipuleucel-T), poly(ADP-ribose) polymerase inhibitors, and radiopharmaceutical therapies.17,18 Treatment is usually tailored based on disease characteristics; patient characteristics, including co-morbidity status, mutational profile, and prior treatment; and physician and patient preference.18 Regardless of strategy and sequence chosen, the goal is to improve overall survival (OS) while minimizing treatment-induced toxicities and maintaining the health-related quality of life of patients during the evolving disease course. Combining therapies uses complementary mechanisms to enhance efficacy, delay resistance, and improve survival. Radium-223 targets bone metastases, while ARPIs and immunotherapies (eg, sipuleucel-T) address systemic disease and soft tissue progression. This multimodal approach seeks to optimize disease control, reduce skeletal-related events, and extend OS.
This review critically examines the mechanisms, clinical applications, safety, and challenges associated with Ra-223 treatment while discussing its potential in combination therapies and ongoing research directions.
Abbreviations
177Lu-PSMA leutetium-177 vipivotide tetraxetan prostate-specific membrane antigen therapy
aBSI automated bone scan index
ADT androgen-deprivation therapy
ARPI androgen receptor pathway inhibitor
HR hazard ratio
IR interleukin
mCRPC metastatic castration-resistant prostate cancer
mHSPC metastatic hormone-sensitive prostate cancer
OS overall survival
PFS progression-free survival
Ra-223 radium-223 dichloride
RT radiation therapy
Methods
To summarize clinical evidence supporting the use of Ra-223, we explored literature, including randomized trials, prospective nonrandomized studies, and large real-world registry-based studies. Prespecified subgroup analyses from prospective studies were also included. Accepting slight differences in end point definitions across trials, the outcome of interests was OS (where reported) and radiographic progression-free survival (PFS), clinician-reported and patient-reported toxicities, and quality of life ( in addition to exploratory end points as appropriate.
Mechanism of Action
Radium-223 is a divalent cation (similar to calcium) that selectively targets areas of osteoblastic activity in bone metastases (Table 1). The bone specificity of Ra-223 is a result of its valence electron configuration, by virtue of which it gets incorporated into areas of increased bone turnover. Radium-223 decays to radon-219, which over 5 more stages decays to lead-207. During the overall process, for each atom of Ra-223, 4 α-particles are released, which represents approximately 95% of the total radiation energy emitted. The α-particles are characterized by their high linear energy transfer and short range. These unique properties enable Ra-223 to deliver potent, localized radiation with minimal off-target spillage. Radium-223 has a half-life of 11.4 days, with α-particle energy of 27.4 MeV, a specific activity of 1.9 MBq/ng, and an α-particle penetration of less than 0.1 mm.19

High Linear Energy Transfer
DNA damage is thought to be the primary mechanism of radionuclide-induced cell death, and linear energy transfer determines the biological effectiveness of the radionuclide therapy. α-Particles are characterized by 400-fold greater linear energy transfer than β-particles. Several hundred β-particle traversals through the nucleus are needed to induce cell death, which could be similarly induced by less than 10 α-decays.20 α-Particles emitted by Ra-223 have a linear energy transfer of approximately 80 keV/μm. This high linear energy transfer causes dense ionization along the particle’s path, resulting in double-stranded DNA breaks in tumor cells. These breaks are difficult for cells to repair, leading to apoptosis and cell death. The high linear energy transfer ensures that a single α-particle has the potential to kill a cell, making α-emitters like Ra-223 highly efficient in targeting cancerous lesions. The α-particles are furthermore less susceptible to hypoxia. This trait contrasts with oxygen-dependent DNA damage caused by external beam RT or activation signaling pathways used in ARPIs and chemotherapeutic agents, which are also oxygen dependent.21 Finally, the cell killing by Ra-223 is also independent of cell cycle status and dose rate.22
Short Range
The range of α-particles is less than 100 μm, corresponding to a few cell diameters.23 This limited range confines the radiation to the immediate vicinity of bone metastases, sparing the surrounding normal tissues and minimizing systemic toxicity. This property is particularly beneficial for patients with mCRPC, who are often frail and unable to tolerate widespread radiation exposure. The biodistribution, pharmacokinetics, and dosimetry of Ra-223 were studied in a phase 1 trial in which a dose of 100 kBq/kg was administered to 6 patients treated twice, 6 weeks apart. The study showed that the gut was the main route of excretion and simultaneously confirmed rapid reduction in radioactivity from the blood. The bone surfaces were observed to receive the greatest absorbed dose. Of note, the range of absorbed doses delivered to bone surfaces was extremely large, ranging between 2.3 Gy/MBq and 13.1 Gy/MBq from α-emissions and between 9 mGy/MBq and 51 mGy/MBq from β emissions and γ emissions. The biodistribution and pharmacokinetic profiles were similar for both administrations. The ranges of absorbed doses delivered to the red marrow were 177 mGy/MBq to 994 mGy/MBq from activity on the bone surfaces and 1 mGy/MBq to 5 mGy/MBq from activity in the blood. No activity-limiting toxicity was observed at these levels of administration. A lack of either severe gastrointestinal toxicity or severe myelotoxicity was assumed to be the result of the very short path length of the α-particles that did not uniformly irradiate the intestinal walls from the gut contents or the marrow from the bone surfaces.24 In another phase 1 pharmacokinetic and biodistribution study, Carrasquillo et al25 conducted a stepwise activity escalation study of Ra-223 at 50 kBq/kg, 100 kBq/kg, and 200 kBq/kg doses in 10 patients. The investigators found a rapid clearance of the radioisotope, with fecal excretion as the major route of elimination, with low levels of urinary excretion.25
Cytotoxicity and Bone Microenvironment Modulation
In addition to its direct cytotoxic effects on tumor cells, Ra-223 exerts substantial effects on the bone microenvironment, and it stabilizes bone architecture. These effects are mediated by different underlying mechanisms.
Radium-223 has been shown to forestall bone metastases while preserving bone structure in prostate cancer xenograft models using lymph node carcinoma of the prostate (androgen-sensitive) cell lines and LuCaP 58 (abiraterone-resistant) cell lines.14 Mice with either cell line were given Ra-223 based on serum prostate-specific antigen levels, and the effect of Ra-223 treatment on several bone and tumor parameters was evaluated. Radium-223 reduced osteoblastic bone growth while preserving bone architecture and bone volume.26
Preclinical studies suggest that Ra-223 reduces angiogenesis within the bone microenvironment, thereby limiting the vascular supply to metastatic tumors. In a clinical study involving 22 patients with prostate cancer with bone metastasis, higher baseline levels of specific cytokines, such as interleukin (IL) 6, were strongly associated with OS. Specifically, IL-6 levels above the median were associated with an inferior OS compared with IL-6 levels below the median. The angiogenic biomarker placental growth factor furthermore inversely correlated with OS, supporting a potential relationship between Ra-223 and antiangiogenic effects.27
Immune Modulation by α-Emitters
Radium-223 and other α-emitters have been shown to interact with the immune system in several ways. Localized tumor cell death induced by Ra-223 can release tumor-associated antigens, potentially priming the immune system for a broader antitumor response.28 The α-particles could trigger immunogenic cell death that primes the immune system through the release of immunostimulatory damage-associated molecular patterns and of high mobility group box 1 proteins, activators of dendritic cells and T cells.29 Lower doses of radiation elicit sublethal DNA damage that is detected by the sensor cyclic GMP-AMP synthase and that leads to the production of type I interferon. Type I interferon may help prime tumor antigen–specific T cells and mount an adaptive response, which can lead to local and distant tumor cell killing.28,29 Cells receiving a sublethal dose of radiation undergo phenotypic changes such as increased expression of major histocompatibility complex, class 1, on the surface of cancer cells that may alter their susceptibility to immune response.28
By targeting areas of active bone remodeling, Ra-223 may disrupt immunosuppressive signals from the tumor microenvironment. Radium-223 could also potentially enhance immune cell infiltration. A study involving 15 patients with mCRPC found that Ra-223 treatment resulted in a clinically significant decrease in the mean frequency of programmed cell death 1 protein, expressing effector memory CD8-positive T cells. The frequency of programmed cell death 1 protein–positive effector memory CD8-positive T cells dropped from 21% before treatment to 15% after 1 treatment course (P = .02). This phenomenon suggests that Ra-223 may enhance T-cell function by decreasing inhibitory signaling.30
The findings from clinical trials that combined Ra-223 with immunotherapy, however, have been inconclusive. In a randomized phase 2 study, the combination of Ra-223 and pembrolizumab was compared with Ra-223 alone in patients with mCRPC. The primary end point, involving differences in CD4-positive and CD8-positive T-cell infiltrates before and after treatment, showed no statistically significant improvement between the combination therapy arm and the Ra-223 treatment groups. For CD4-positive T cells, the median fold-change was −0.7 for the combination therapy group and 0.1 for the Ra-223 group (P = .66). For CD8-positive T cells, the fold-change was −0.6 with combination therapy and −1.3 with Ra-223 alone (P = .66), indicating minimal impact on T-cell infiltration.31
In another small study, 32 patients were randomly assigned to treatment with sipuleucel-T vs 6 cycles of Ra-223 with interdigitated sipuleucel-T. Patients in the sipuleucel-T alone group exhibited a 3.2-fold greater change in T-cell proliferation compared with patients receiving the combination therapy. Although both groups showed increased absolute antibody responses to target antigens (PA2024 and prostatic acid phosphatase), there were no statistically significant differences between the groups in terms of the magnitude of response. Likewise, cytokine analyses revealed no statistically significant changes in key immune mediators between the treatment arms. Despite the lower immune responses observed with combination therapy, clinical outcomes suggested substantial efficacy. Notably, the combination arm showed a higher proportion of patients achieving a decline in prostate-specific antigen levels greater than 50% (31% vs 0%) and significantly improved PFS compared with patients in the sipuleucel-T alone arm.32 This result suggests that there could be potential mechanisms by which Ra-223 and sipuleucel-T synergize, a hypothesis that requires further exploration.
Evidence Favoring the Use of Ra-223 in Prostate Cancer
The first in-human experience with Ra-223 was reported in a single-arm phase 1 trial of 25 patients with metastatic prostate or breast cancer and bone metastases. Remarkably mild hematologic toxicity was observed. Grade 3 neutropenia occurred in 2 patients, and leukopenia occurred in 3 patients. Pain relief was reported by 52% of patients after 7 days, 60% of patients after 4 weeks, and 56% of patients after 8 weeks. Median survival was more than 20 months.33 Given these findings, studies continued to explore the utility of Ra-223 in patients with metastatic prostate cancer, the most osteotropic of all malignancies. Among these studies, a randomized phase 2 trial enrolled 64 patients with mCRPC who required palliative external beam RT for their skeletal metastasis. Patients were assigned to either 4 cycles of Ra-223 (50 kBq/kg) or placebo. Although not a primary end point of the study, it was striking that OS was significantly better for patients in the Ra-223 arm after adjustment for baseline covariates. As expected, time to skeletal-related events was significantly prolonged in the Ra-223 group, with no statistically significant differences in hematologic toxicity.
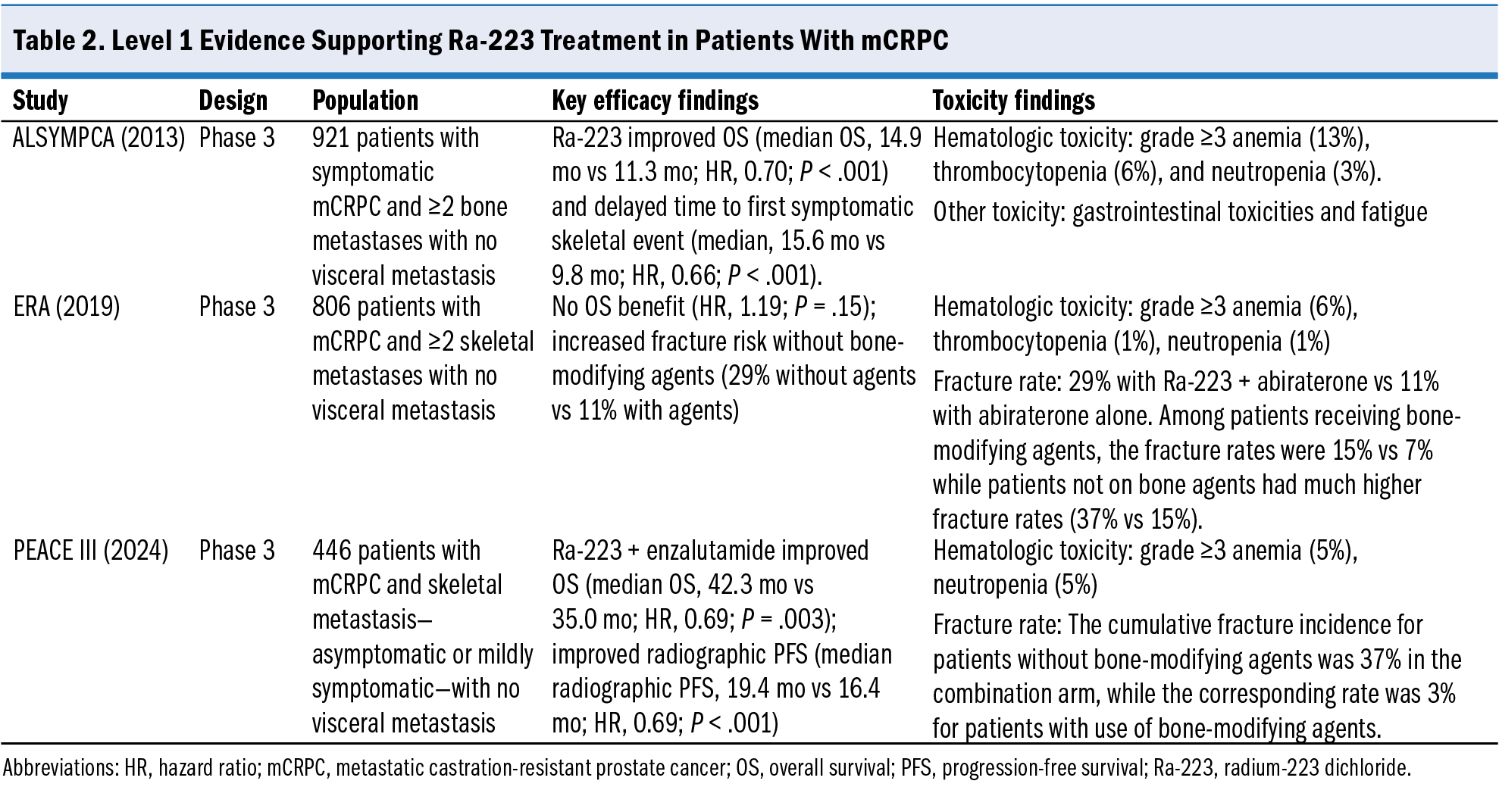
Evidence in mCRPC
Radium-223’s efficacy in mCRPC has been extensively studied in multiple trials, establishing it as a cornerstone of disease management for patients with mCRPC and symptomatic bone metastases. The pivotal study that established Ra-223 as a treatment option for mCRPC is the ALSYMPCA trial (Table 2). This phase 3 randomized, double-blind, placebo-controlled trial evaluated the efficacy and safety of Ra-223 in men with symptomatic mCRPC and bone metastases. A total of 921 patients were assigned to receive 6 cycles of Ra-223 (50 kBq/kg) or matching placebo, with the primary end point of OS.5 The results demonstrated a statistically significant improvement in OS for patients treated with Ra-223 compared with patients receiving placebo, with median survival times of 14.9 months vs 11.3 months, respectively, which translated into a 30% reduction in the risk for death (hazard ratio [HR], 0.70 [95% CI, 0.58-0.83]; P < .001). Patients in the Ra-223 arm also demonstrated longer time to first symptomatic skeletal event (median, 15.6 vs 9.8 months; HR, 0.66 [95% CI, 0.52-0.83]; P < .001) than patients in the placebo arm.5 The serious adverse events in the Ra-223 group included bone pain, anemia, and spinal cord compression. There was only 1 incident of a grade 5 thrombocytopenia event in the experimental arm, possibly related to the study drug. A significantly higher percentage of patients receiving Ra-223 experienced meaningful improvement in EuroQol-5 Dimension utility score on treatment vs placebo (29.2% vs 18.5%, respectively; odds ratio, 1.82 [95% CI, 1.21-2.74]).34 A near-contemporary phase 3b study reported the safety and efficacy of concomitant enzalutamide or abiraterone with Ra-223 in which 58% of patients completed all 6 cycles of Ra-223. Among patients who were given at least 1 dose of Ra-223, 58% experienced treatment-related adverse events. The most common grade 3 or higher adverse events were anemia (5%) and thrombocytopenia (2%), and median OS was 16 months (95% CI, 13-not reached).35
Dosage of Ra-223 was investigated in another phase 2 trial in which patients were randomly assigned to 6 cycles of a standard dose (55 kBq/kg every 4 weeks), a high dose (88 kBq/kg every 4 weeks), or an extended dose (55 kBq/kg every 4 weeks for 12 cycles) of Ra-223. There was no statistically significant difference in symptomatic skeletal event–free survival among the 3 groups with similar OS rates. The treatment-emergent adverse event rates were furthermore significantly higher in the high-dose and extended-dose arms. Six cycles of standard-dose Ra-223 therefore continue to be the appropriate treatment regimen for men with mCRPC.36
The safety and tolerability of ARPIs (abiraterone or enzalutamide) with concurrent Ra-223 were evaluated in 2 phase 2 trials (ERADICATE and ENZARADICATE) in patients with symptomatic mCRPC and bone metastases. In both studies, these combination regimens demonstrated some clinical benefit without any undue increase in the risk for toxicity.37,38
Subsequently, the ERA 223 trial investigated the efficacy of Ra-223 in conjunction with abiraterone for chemotherapy-naive patients with asymptomatic or mildly symptomatic mCRPC and osseous metastases (Table 2).39 Overall, 806 patients were randomly assigned to receive Ra-223 or placebo in addition to abiraterone plus prednisone. The authors found a median symptomatic skeletal event–free survival of 22.3 months in the Ra-223 group and 26.0 months in the placebo group (HR, 1.12 [95% CI, 0.92-1.37]). There was no difference in the radiographic PFS rates (11.2 months vs 12.4 months; HR, 1.15 [95% CI, 0.96-1.38]) or OS rates (HR, 1.19 [95% CI, 0.95-1.51]) between the 2 arms. Serious treatment-related adverse events occurred in 41% of patients in the Ra-223 group and 39% of patients in the placebo group. Fractures occurred in 29% of patients receiving Ra-223 compared with 11% of patients in the abiraterone-only arm (HR, 3.13 [95% CI, 2.21-4.45]). Osteoporotic fractures were the most common type of fracture in the Ra-223 group, and of all fracture types, they differed the most between the study arms. Of note, the use of bone-modifying agents was not mandated in this trial. Only 39% of patients in the combination arm and 42% of patients in the placebo arm received them. In a post hoc analysis, among patients who used bone-modifying agents, 15% of patients in the Ra-223 arm and 7% of patients in the placebo arm experienced a fracture, while the corresponding fracture rates of patients not taking bone-modifying agents were significantly higher at 37% in the Ra-223 arm and 15% in the placebo arm. These findings highlight the critical importance of optimizing supportive care such as bone-modifying agents when integrating Ra-223 into combination regimens.
The PEACE III trial investigated Ra-223 in combination with enzalutamide (Table 2). It was a phase 3 study that enrolled 426 patients across 12 countries from 2015 to 2023. Eligible patients had CRPC with bone metastases; were asymptomatic or mildly symptomatic; and had not received prior treatment with enzalutamide, apalutamide, darolutamide, or Ra-223. Patients were randomly assigned 1:1 to receive either 160 mg enzalutamide once daily (standard of care) or 160 mg enzalutamide once daily plus Ra-223 every 4 weeks for 6 cycles.40 To address the fracture risk noted in ERA 223, the trial protocol for PEACE III was amended to mandate the use of bone-modifying agents (denosumab and zoledronic acid) and baseline dual-energy x-ray absorptiometry scans. This amendment led to an increase in the use of bone-modifying agents, from 46% to 97%.41 The combination improved radiographic PFS (median radiographic PFS, 19.4 months vs 16.4 months; HR, 0.69 [95% CI, 0.54-0.87]; P < .001) and OS (median OS, 42.3 months vs 35.0 months; HR, 0.69 [95% CI, 0.52-0.90]; P = .003). Time to next systemic treatment was also improved in the combination arm, with only 30% of patients in this treatment arm requiring a next-line systemic treatment at 24 months of follow-up compared with 51% of patients in the enzalutamide monotherapy arm (HR, 0.57 [95% CI, 0.44-0.75]; P < .001). In contrast to the ERA 223 trial, there were no statistically significant differences in time to skeletal-related events, with 18% of patients in both arms having a skeletal-related event by 24 months of follow-up. The most frequent grade 3 or higher treatment-emergent adverse events in the combination arm were hypertension (34%), fatigue (6%), anemia (5%), and neutropenia (5%). After 1 year of treatment, the cumulative fracture incidence for patients without bone-modifying agents was 37% in the combination arm, while the corresponding rate was 3% for patients with use of bone-modifying agents. This trial confirmed that Ra-223 can be safely combined with ARPIs when adequate bone health measures are employed.
The combination of Ra-223 with other anticancer interventions, such as docetaxel, will become clearer with the results of the DORA trial (ClinicalTrials.gov identifier NCT03574571), which is an ongoing phase 3 study evaluating Ra-223 use in combination with docetaxel for patients with mCRPC and extensive bone metastases. Although the trial has not yet reported its findings, the results are highly anticipated because they could establish Ra-223 as a synergistic partner with chemotherapy in high-burden disease.
Real-world studies outside clinical trials have reported the safety and efficacy of Ra-223. In the RAPIT study by Huang et al,42 Ra-223 was first-line therapy in 23.2% of patients and second-line therapy in 47.7% of patients. Approximately 84% of patients with first-line use of Ra-223 and 66% of patients with second-line use completed 5 to 6 cycles of Ra-223. Any grade of treatment-emergent adverse events occurred in 54% of patients (serious adverse events in 28.6% of patients), with 2.2% being fractures.42 Median OS was 16 months (95% CI, 12.1-19.5 months), which was congruent with clinical trial data. The findings were similar to those of REASSURE, another real-world observational study of Ra-223 use in patients with mCRPC. Concomitant enzalutamide was received by 31% of patients, and 47% of patients received concomitant bone health agents. Any grade of drug-related treatment-emergent adverse events occurred in 32% of patients, and grade 3 or higher drug-related treatment-emergent adverse events occurred in 10% of patients. Overall, 4% of patients had fractures, and 2% of patients developed bone disorders. Median OS was 17.8 months (95% CI, 15.6-19.4 months).43 In another report from the REASSURE study, 182 patients received taxane-based chemotherapy after Ra-223, and only 2 of those patients had severe neutropenia during chemotherapy. Median OS was 24.3 months from Ra-223 initiation and 11.8 months from the start of taxane therapy.44 Another small study with 49 patients (RALU) furthermore showed that use of Ra-223 therapy in patients with mCRPC does not obviate the possibility of using 177Lu-PSMA therapy.45 Most common grade 3 or higher adverse events were hematologic (anemia in 18% of patients and thrombocytopenia in 2% of patients). Median OS from the start of Ra-223 was 31 months.
Evidence in mHSPC
The role of Ra-223 in mHSPC is emerging, with pivotal insights provided by the ADRAD trial46 and additional findings from the RAVENS trial47 along with other single-institution studies.
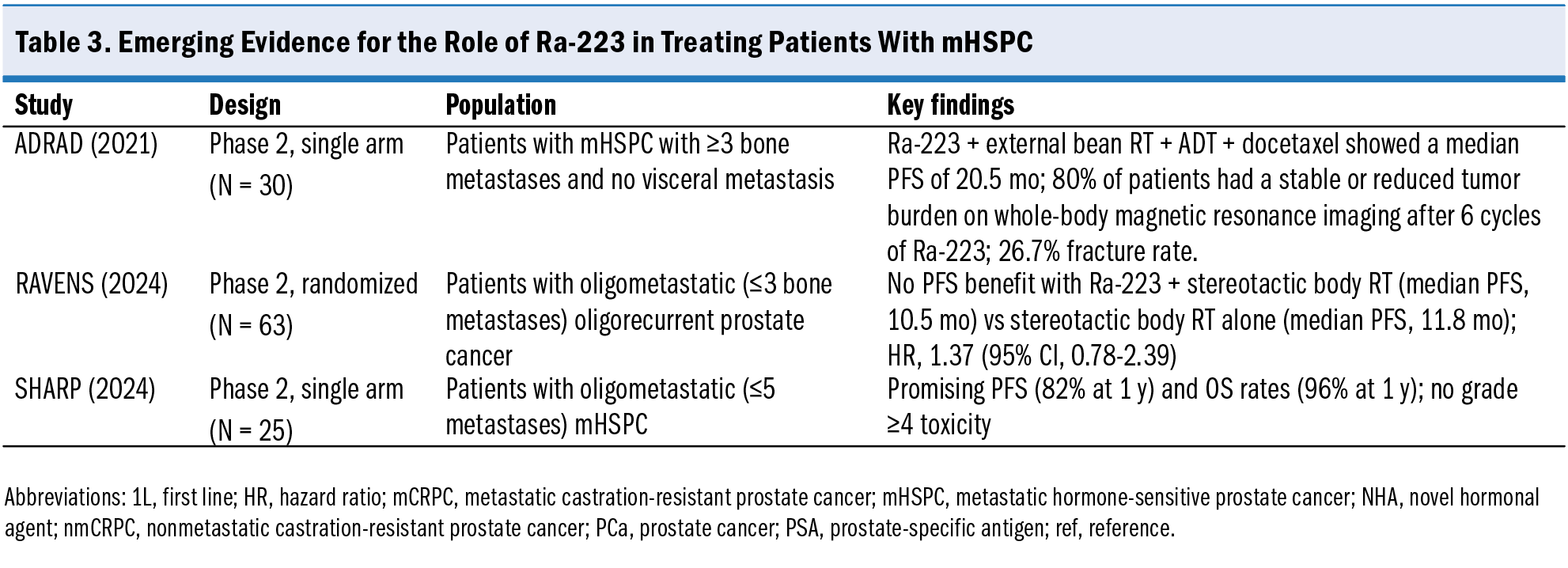
The ADRAD trial evaluated the integration of Ra-223 with external beam RT, ADT, and 6 cycles of docetaxel in patients with de novo mHSPC with at least 3 bone metastases and no visceral metastases (Table 3).46 Patients were treated with a combination of external beam RT and Ra-223 for the first 2 months, then Ra-223 only for the subsequent 4 months of treatment. Patients treated with Ra-223 achieved a median PFS of 20.5 months. Imaging assessments demonstrated that 80% of patients had a stable or reduced tumor burden on whole-body magnetic resonance imaging after 6 cycles of Ra-223. By the end of the study, stable or reduced tumor burden was maintained in 56% of patients. Approximately 90% of patients exhibited reductions in alkaline phosphatase, reflecting robust activity against bone metastases. The alkaline phosphatase decline, along with reduction in tumor burden on magnetic resonance imaging, was evident in a subgroup of patients beyond 1 year after docetaxel completion. This observation suggests that certain patients derived anticancer benefit from the combination of Ra-223 and external beam RT beyond the effect of systemic therapy. Approximately 26.7% of patients, however, experienced fractures. Hematologic toxicities were mild, with grade 3 leukopenia in 10% of patients and thrombocytopenia in 3.3% of patients. A biomarker substudy revealed that patients with late relapses had higher levels of DNA damage in circulating lymphocytes along with a decrease in hemoglobin and red blood cell counts relative to patients with early relapses.48 Increases in DNA damage during the Ra-223–only cycles of treatment were specifically related to the response in these patients. These findings underscore the potential of radiosensitivity markers to refine patient selection and optimize outcomes.
The RAVENS trial evaluated the addition of Ra-223 to stereotactic body RT in recurrent oligometastatic (ie, ≤3 metastases and no visceral disease) prostate cancer (Table 3). Patients receiving Ra-223 with stereotactic body RT (n = 30) had a median PFS of 10.5 months compared with a median PFS of 11.8 months in patients treated with stereotactic body RT alone (HR, 1.37 [95% CI, 0.78-2.39]).47 There was no difference in metastasis-free survival or ADT-free survival. One of the potential reasons for the limited efficacy of this combination could be lack of spatial synergism between the 2 modalities, especially in the context of limited metastatic disease burden. Both Ra-223 and stereotactic body RT in this trial predominantly targeted bone metastases and thus failed to stall the emergence of disease failure.
In SHARP, another phase 2 single-institution trial,49 Ra-223 was combined with 36 weeks of ADT and stereotactic body RT for 25 patients with oligometastatic (≤5 metastases) mHSPC (Table 3). In addition, patients with de novo mHSPC received prostate radiation with or without pelvic radiation. Patients received 6 infusions of Ra-223 starting 1 month after stereotactic body RT. Median time to treatment failure in the entire study cohort was 19.0 months, 34.3 months for patients with de novo mHSPC, and 17.7 months for patients with oligoprogressive mHSPC. Although the sample size and follow-up were limited, the survival results are promising. At 1 year, PFS was 82.1% in all patients, 83% in patients with de novo disease, and 81% in patients with oligoprogressive mHSPC. Overall survival at 1 year was 96% in all patients, 100% in patients with de novo disease, and 94% in patients with oligoprogressive mHSPC. No grade 4 or greater toxicities occurred, and 5 patients developed fractures. Interleukin-8 significantly increased from baseline to the end of treatment, and a reduction in proinflammatory IL-1β and tumor necrosis factor-α from baseline was observed.
Biomarkers for Ra-223 Use
Biomarkers are increasingly recognized as critical tools for optimizing Ra-223 therapy because they can predict response and help refine patient selection. The automated bone scan index (aBSI) provides a quantitative assessment of metastatic osseous burden as a percentage of total skeletal weight using advanced imaging. Studies indicate that patients with lower baseline aBSI and minimal increases during treatment derive better outcomes. For example, a baseline aBSI less than 0.9 was associated with a median OS of 39.9 months compared with 15.2 months for an aBSI of at least 0.9 (HR, 5.87; P = .003). Overall survival was significantly longer for participants who had a relatively stable aBSI at 2 months relative to baseline (<0.7 increase) compared with patients with an increase of at least 0.7 (median OS, 36.6 vs 17.9 months; P = .01).50 Circulating tumor cell counts and molecular profiles offer additional prognostic and predictive insights in the context of using Ra-223 to treat patients with Ra-223. High baseline circulating tumor cell levels (≥5/7.5 mL) and baseline expression of androgen receptor splice variant-7 have been shown to portend worse outcomes in patients with mCRPC treated with mCRPC.50 In another small study that randomly assigned 39 patients to treatment with Ra-223 and enzalutamide vs treatment with Ra-223 alone, the authors noted a statistically significant decline in serum bone marrow microenvironment N-telopeptide levels with combination therapy. This decline correlated with superior prostate-specific antigen response.51 Further studies are needed, however, for prospective validation of these emerging biomarkers and to determine their independent or combined use in the context of Ra-223 in advanced prostate cancer.
Conclusions
Radium-223 is a valuable, life-prolonging therapeutic option for patients with mCRPC and bone-predominant disease. Studies have provided robust evidence supporting its ability to prolong survival, delay skeletal-related events, and improve quality of life as a monotherapeutic agent. Recent evidence also supports the utility of Ra-223 in combination with ARPI agents such as enzalutamide. The role of supportive care regimens such as bone-modifying agents, however, are pivotal in the context of such combination therapy. Emerging evidence shows the potential utility of Ra-223 in an mHSPC setting, though we need level I evidence to support the integration of Ra-223 into the current landscape of management in mHSPC. Ongoing studies will be crucial to refining its use in combination regimens, identifying predictive biomarkers, and exploring its potential role in immunomodulation and theranostics.
References
1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394-424. doi:10.3322/caac.21492
2. Albertsen PC, Hanley JA, Fine J. 20-year outcomes following conservative management of clinically localized prostate cancer. JAMA. 2005;293(17):2095-2101. doi:10.1001/jama.293.17.2095
3. Roy S, Morgan SC. Who dies from prostate cancer? An analysis of the Surveillance, Epidemiology and End Results database. Clin Oncol. 2019;31(9):630-636. doi:10.1016/j.clon.2019.04.012
4. Parker CC, James ND, Brawley CD, et al; STAMPEDE Trial Collaborative Group. Radiotherapy to the prostate for men with metastatic prostate cancer in the UK and Switzerland: long-term results from the STAMPEDE randomised controlled trial. PLoS Med. 2022;19(6);e1003998. doi:10.1371/journal.pmed.1003998
5. Parker C, Nilsson S, Heinrich D, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369(3):213-223. doi:10.1056/NEJMoa1213755
6. Francolini G, Gaetano Allegra A, Detti B, et al. Stereotactic body radiation therapy and abiraterone acetate for patients affected by oligometastatic castrate-resistant prostate cancer: a randomized phase II trial (ARTO). J Clin Oncol. 2023;41(36):5561-5568. doi:10.1200/JCO.23.00985
7. Tang C, Sherry AD, Haymaker C, et al. Addition of metastasis-directed therapy to intermittent hormone therapy for oligometastatic prostate cancer: the EXTEND phase 2 randomized clinical trial. JAMA Oncol. 2023;9(6):825-834. doi:10.1001/jamaoncol.2023.0161
8. Phillips R, Shi WY, Deek M, et al. Outcomes of observation vs stereotactic ablative radiation for oligometastatic prostate cancer: the ORIOLE phase 2 randomized clinical trial. JAMA Oncol. 2020;6(5):650-659. doi:10.1001/jamaoncol.2020.0147
9. Sartor O, de Bono J, Chi KN, et al; VISION Investigators. Lutetium-177-PSMA-617 for metastatic castration-resistant prostate cancer. N Engl J Med. 2021;385(12):1091-1103. doi:10.1056/NEJMoa2107322
10. Morris MJ, de Bono JS, Chi KN, et al. Phase III study of lutetium-177-PSMA-617 in patients with metastatic castration-resistant prostate cancer (VISION). J Clin Oncol. 2021;39(18_suppl):LBA4. doi:10.1200/JCO.2021.39.15_suppl.LBA4
11. Devlies W, Eckstein M, Cimadamore A, et al. Clinical actionability of the genomic landscape of metastatic castration resistant prostate cancer. Cells. 2020;9(11):2494. doi:10.3390/cells9112494
12. Barbieri CE, Bangma CH, Bjartell A, et al. The mutational landscape of prostate cancer. Eur Urol. 2013;64(4):567-576. doi:10.1016/j.eururo.2013.05.029
13. Ottewell PD, Wang N, Meek J, et al. Castration-induced bone loss triggers growth of disseminated prostate cancer cells in bone. Endocr Relat Cancer. 2014;21(5):769-781. doi:10.1530/ERC-14-0199
14. Sartor O, Reid RH, Hoskin PJ, et al; Quadramet 424Sm10/11 Study Group. Samarium-153-lexidronam complex for treatment of painful bone metastases in hormone-refractory prostate cancer. Urology. 2004;63(5):940-945. doi:10.1016/j.urology.2004.01.034
15. Tu SM, Delpassand ES, Jones D, Amato RJ, Ellerhorst J, Logothetis CJ. Strontium-89 combined with doxorubicin in the treatment of patients with androgen-independent prostate cancer. Urol Oncol. 1996;2(6):191-197. doi:10.1016/S1078-1439(97)00013-6
16. James ND, Pirrie SJ, Pope AM, et al. Clinical outcomes and survival following treatment of metastatic castrate-refractory prostate cancer with docetaxel alone or with strontium-89, zoledronic acid, or both: the TRAPEZE randomized clinical trial. JAMA Oncol. 2016;2(4):493-499. doi:10.1001/jamaoncol.2015.5570
17. Kulasegaran T, Oliveira N. Metastatic castration-resistant prostate cancer: advances in treatment and symptom management. Curr Treat Options Oncol. 2024;25(7):914-931. doi:10.1007/s11864-024-01215-2
18. Roy S, Saad F. Metastatic castrate-resistant prostate cancer: a new horizon beyond the androgen receptors. Curr Opin Support Palliat Care. 2022;16(4):223-229. doi:10.1097/SPC.0000000000000620
19. Brito AE, Etchebehere E. Radium-223 as an approved modality for treatment of bone metastases. Semin Nucl Med. 2020;50(2):177-192. doi:10.1053/j.semnuclmed.2019.11.005
20. King AP, Lin FI, Escorcia FE. Why bother with alpha particles? Eur J Nucl Med Mol Imaging. 2021;49:7-17. doi:10.1007/s00259-021-05431-y
21. Park SY, Kim YJ, Gao AC, et al. Hypoxia increases androgen receptor activity in prostate cancer cells. Cancer Res. 2006;66(10):5121-5129. doi:10.1158/0008-5472.CAN-05-1341
22. Nilsson S. Alpha-emitter radium-223 in the management of solid tumors: current status and future directions. ASCO Educational Book. 2014;34(1):e132-139. doi:10.14694/EdBook_AM.2014.34.e132
23. Flux GD. Imaging and dosimetry for radium-223: the potential for personalized treatment. Br J Radiol. 2017;90(1077):20160748. doi:10.1259/bjr.20160748
24. Chittenden SJ, Hindorf C, Parker CC, et al. A phase 1, open-label study of the biodistribution, pharmacokinetics, and dosimetry of 223Ra-dichloride in patients with hormone-refractory prostate cancer and skeletal metastases. J Nucl Med. 2015;56(9):1304-1309. doi:10.2967/jnumed.115.157123
25. Carrasquillo JA, O’Donoghue JA, Pandit-Taskar N, et al. Phase I pharmacokinetic and biodistribution study with escalating doses of 223Ra-dichloride in men with castration-resistant metastatic prostate cancer. Eur J Nucl Med Mol Imaging. 2013;40:1384-1393. doi:10.1007/s00259-013-2427-6
26. Suominen MI, Fagerlund KM, Rissanen JP, et al. Radium-223 inhibits osseous prostate cancer growth by dual targeting of cancer cells and bone microenvironment in mouse models. Clin Cancer Res. 2017;23(15):4335-4346. doi:10.1158/1078-0432.CCR-16-2955
27. Saylor PJ, Kozin SV, Matsui A, et al. The radiopharmaceutical radium-223 has immunomodulatory effects in patients and facilitates anti-programmed death receptor-1 therapy in murine models of bone metastatic prostate cancer. Radiother Oncol. 2024;192:110091. doi:10.1016/j.radonc.2024.110091
28. Malamas AS, Gameiro SR, Knudson KM, Hodge JW. Sublethal exposure to alpha radiation (223Ra dichloride) enhances various carcinomas’ sensitivity to lysis by antigen-specific cytotoxic T lymphocytes through calreticulin-mediated immunogenic modulation. Oncotarget. 2016;7:86937-86947. doi:10.18632/oncotarget.13520
29. Golden EB, Frances D, Pellicciotta I, Demaria S, Barcellos-Hoff MH, Formenti SC. Radiation fosters dose-dependent and chemotherapy-induced immunogenic cell death. Oncoimmunology. 2014;3:e28518. doi:10.4161/onci.28518
30. Kim JW, Shin MS, Kang Y, Kang I, Petrylak DP. Immune analysis of radium-223 in patients with metastatic prostate cancer. Clin Genitourin Cancer. 2017;16(2):e469-e476. doi:10.1016/j.clgc.2017.10.010
31. Choudhury AD, Kwak L, Cheung A, et al. Randomized phase II study evaluating the addition of pembrolizumab to radium-223 in metastatic castration-resistant prostate cancer. Cancer Immunol Res. 2024;12(6):704-718. doi:10.1158/2326-6066.CIR-22-0306
32. Marshall CH, Fu W, Wang H, et al. Randomized phase II trial of sipuleucel-T with or without radium-223 in men with bone-metastatic castration-resistant prostate cancer. Clin Cancer Res. 2021;27(6):1623-30. doi:10.1158/1078-0432.CCR-20-4476
33. Nilsson S, Larsen RH, Fosså SD, et al. First clinical experience with alpha-emitting radium-223 in the treatment of skeletal metastases. Clin Cancer Res. 2005;11(12):4451-4459. doi:10.1158/1078-0432.CCR-04-2244
34. Nilsson S, Cislo P, Sartor O, et al. Patient-reported quality-of-life analysis of radium-223 dichloride from the phase III ALSYMPCA study. Ann Oncol. 2016;27(5):868-874. doi:10.1093/annonc/mdw065
35. Saad F, Carles J, Gillessen S, et al. Radium-223 and concomitant therapies in patients with metastatic castration-resistant prostate cancer: an international, early access, open-label, single-arm phase 3b trial. Lancet Oncol. 2016;17(9):1306-1316. doi:10.1016/S1470-2045(16)30173-5
36. Sternberg CN, Saad F, Graff JN, et al. A randomised phase II trial of three dosing regimens of radium-223 in patients with bone metastatic castration-resistant prostate cancer. Ann Oncol. 2020;31(2):257-265. doi:10.1016/j.annonc.2019.10.025
37. Shore ND, Schellhammer PF, Tutrone RF, Mariados NF, Harrelson SS. Open label phase II study of enzalutamide with concurrent administration of radium 223 dichloride in patients with castration-resistant prostate cancer. Clin Genitourin Cancer. 2020;18(5):416-422. doi:10.1016/j.clgc.2020.02.015
38. Shore ND, Tutrone RF, Mariados NF, et al. eRADicAte: a prospective evaluation combining radium-223 dichloride and abiraterone acetate plus prednisone in patients with castration-resistant prostate cancer. Clin Genitourin Cancer. 2018;16(2):149-154. doi:10.1016/j.clgc.2017.10.022
39. Smith M, Parker C, Saad F, et al. Addition of radium-223 to abiraterone acetate and prednisone or prednisolone in patients with castration-resistant prostate cancer and bone metastases (ERA 223): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(3):408-419. doi:10.1016/S1470-2045(18)30860-X
40. Gillessen S, Choudhury A, Saad F, et al. LBA1 A randomized multicenter open label phase III trial comparing enzalutamide vs a combination of radium-223 (Ra223) and enzalutamide in asymptomatic or mildly symptomatic patients with bone metastatic castration-resistant prostate cancer (mCRPC): first results of EORTC-GUCG 1333/PEACE-3. Ann Oncol. 2024;35(suppl 2):S1254. doi:10.1016/j.annonc.2024.08.2307
41. Gillessen S, Tombal B, Turco F, et al. Decrease in fracture rate with mandatory bone-protecting agents in the EORTC 1333/PEACE-3 trial comparing radium-223 combined with enzalutamide versus enzalutamide alone: a safety analysis. Eur Urol. 2025;87(3):285-288. doi:10.1016/j.eururo.2024.11.027
42. Huang CY, Huang CP, Huang YY, et al. Real-world safety and effectiveness of radium-223 in patients with metastatic castration-resistant prostate cancer: interim analyses of the prospective, observational RAPIT study. Int J Cancer. 2024;155(7):1268-1277. doi:10.1002/ijc.35040
43. Song DY, George S, Zimberg S, et al. Real-world safety and effectiveness of radium-223 (223Ra) in patients (pts) with metastatic castration-resistant prostate cancer (mCRPC) treated in the US: the non-interventional REASSURE study. J Clin Oncol. 2023;41(16_suppl):5050. doi:10.1200/JCO.2023.41.16_suppl.5050
44. Higano CS, Dizdarevic S, Logue J, et al. Safety and effectiveness of the radium-223-taxane treatment sequence in patients with metastatic castration-resistant prostate cancer in a global observational study (REASSURE). Cancer. 2024;130(11):1930-1939. doi:10.1002/cncr.35221
45. Rahbar K, Essler M, Pabst KM, et al. Safety and survival outcomes of 177Lu-prostate-specific membrane antigen therapy in patients with metastatic castration-resistant prostate cancer with prior 223Ra treatment: the RALU study. J Nucl Med. 2023;64(4):574-578. doi:10.2967/jnumed.122.264456
46. Turner PG, Jain S, Cole A, et al. Toxicity and efficacy of concurrent androgen deprivation therapy, pelvic radiotherapy, and radium-223 in patients with de novo metastatic hormone-sensitive prostate cancer. Clin Cancer Res. 2021;27(16):4549-4556. doi:10.1158/1078-0432.CCR-21-0685
47. Tran PT, Sherry AD, Bazyar S, et al. Outcomes of RAdium-223 and SABR vs SABR for oligomEtastatic prostate caNcerS—the RAVENS phase II randomized trial. Int J Radiat Oncol Biol Phys. 2024;120(2_suppl):S7. doi:10.1016/j.ijrobp.2024.08.021
48. Redmond KM, Turner PG, Cole A, Jain S, Prise KM, O’Sullivan JM. A potential biomarker of radiosensitivity in metastatic hormone sensitive prostate cancer patients treated with combination external beam radiotherapy and radium-223. Radiother Oncol. 2024;191:110063. doi:10.1016/j.radonc.2023.110063
49. Dandapani SV, Hao C, Ladbury CJ, et al. Initial results of a phase 2 trial of stereotactic body radiation therapy, hormone/androgen deprivation therapy and radium 223 dichloride for oligometastatic castrate sensitive prostate cancer (SHARP). Int J Radiat Oncol Biol Phys. 2024;120(suppl 2):S147-S148. doi:10.1016/j.ijrobp.2024.07.2157
50. Saylor PJ, Otani K, Balza R, et al. Circulating and imaging biomarkers of radium-223 response in metastatic castration-resistant prostate cancer. JCO Precis Oncol. 2024;8:e2300230. doi:10.1200/PO.23.00230
51. Agarwal N, Nussenzveig R, Hahn AW, et al. Prospective evaluation of bone metabolic markers as surrogate markers of response to radium-223 therapy in metastatic castration-resistant prostate cancer. Clin Cancer Res. 2020;26(9):2104-2110. doi:10.1158/1078-0432.CCR-19-2591
Article Information
Published: 3/31/2025.
Conflict of Interest Disclosures: Dr Roy reports a Young Investigator Award from the Prostate Cancer Foundation. In addition, Dr Roy reports a research grant from Swim Across America and honorarium from Varian. Dr Spratt reports personal fees from Astellas, AstraZeneca, Bayer, Boston Scientific, GSK, Janssen, Novartis, and Pfizer not related to this work. Dr Barata reports grants and personal fees from Astellas; AstraZeneca; Bayer; Guardant Health; Bristol Myers Squibb; Caris Life Sciences; Dendreon; Eisai; EMD Serono; ESSA Pharma; Exelixis; Ipsen; Janssen; Merck; Merus; Myovant; OncLive; Pfizer, Seattle Genetics; Targeted Oncology; and UroToday not related to this work.
Funding/Support: None.
Author Contributions: Drs Spratt and Roy were responsible for the article concept and design. Mr Roy and Drs Spratt, Barata, and Jia were responsible for review and supervision.
Data Availability Statement: None.