Introduction
The therapeutic landscape for men with metastatic hormone-sensitive prostate cancer (mHSPC) has evolved in recent years. First-line standard of care now consists of systemic therapy with docetaxel and androgen-deprivation therapy (ADT), doublet therapy that includes androgen receptor axis–targeted therapies (ARATs) (eg, abiraterone acetate plus prednisolone [AAP], enzalutamide, or apalutamide [APA]) with ADT), or triple therapy consisting of an ARAT agent, docetaxel, and ADT combination treatment. More recently, darolutamide triple therapy has been approved for the treatment of adult men with mHSPC by the US Food and Drug Administration,1 the European Medicines Agency,2 and multiple health authorities. Evidence from the Darolutamide in Addition to Standard Androgen Deprivation Therapy and Docetaxel in Metastatic Hormone-Sensitive Prostate Cancer (ARASENS) phase 3 trial (ClinicalTrials.gov identifier NCT02799602) showed a significant improvement in overall survival (OS) with darolutamide triple therapy compared with docetaxel plus ADT.3
Key Points
The article reviews and compares published ITCs and meta-analyses in the treatment of patients with mHSPC that include darolutamide triple therapy, identifying inconsistencies and discussing limitations in the process.
We assessed published evidence and described differences in trials as well as the need to standardize methodology for PFS and safety end points to ensure robustness and interpretability of findings.
Indirect treatment comparisons showed a higher ranking with darolutamide triple therapy in terms of OS compared with other treatment options despite methodological variability and imprecise results.
Abbreviations
AAP abiraterone acetate plus prednisone
ADT androgen-deprivation therapy
AE adverse event
APA apalutamide
ARASENS Darolutamide in Addition to Standard Androgen Deprivation Therapy and Docetaxel in Metastatic Hormone-Sensitive Prostate Cancer
ARAT androgen receptor axis–targeted therapy
CINeMA Confidence in Network Meta-Analysis
ENZAMET Enzalutamide in First Line Androgen Deprivation Therapy for Metastatic Prostate Cance
GRADEGrading of Recommendations Assessment, Development and Evaluation
HRhazard ratio
HRQOLhealth-related quality of life
IPDindividual patient data
ITCindirect treatment comparison
mHSPCmetastatic hormone-sensitive prostate cancer
OSoverall survival
PFSprogression-free survival
PRISMAPreferred Reporting Items for Systematic Reviews and Meta-Analyses
PSperformance score
STAMPEDESystemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation of Drug Efficacy
Indirect treatment comparisons (ITCs) allow treatments to be compared in the absence of or owing to insufficient evidence from head-to-head trials and are often conducted using network meta-analysis. The validity of ITCs depends on the studies on which they are based because of basic assumptions about homogeneity. Many ITCs have compared the efficacy and safety of treatment alternatives in mHSPC that have not been compared in head-to-head trials.4 Fisher et al4 highlighted variations in eligibility criteria, including data and statistical methodologies, and reported inadequacies across trials in the treatment of advanced prostate cancer. Given the variety and increasing number of publications in this field, we systematically identified and summarized the findings of published meta-analyses (including ITCs) that include findings for darolutamide triple therapy from the ARASENS trial.3 We aimed to determine whether findings from these studies were consistent, and we discuss both limitations and possible improvements.
Methods
The study protocol was prospectively registered with PROSPERO (CRD42023429478).
Search Strategy
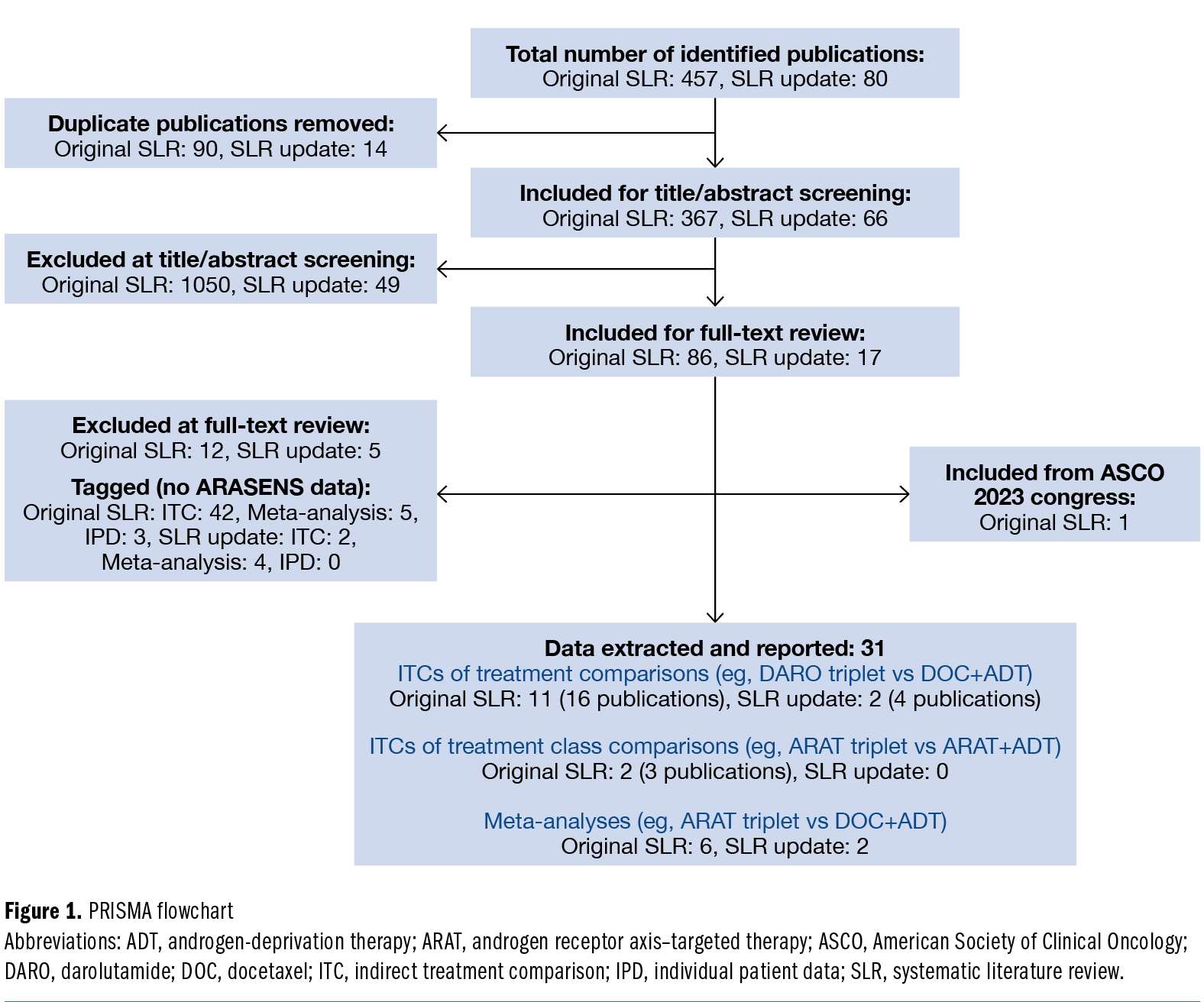
This systematic review was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guidelines. A search was conducted of the MEDLINE, Embase, and Cochrane databases to identify ITCs and meta-analyses published through February 12, 2024 (Supplementary Table 1). Although no restrictions to search dates were applied, only ITCs and meta-analyses that included the ARASENS trial results were considered. This review therefore focuses on the latest trial results in mHSPC. Studies that met the eligibility criteria but that were published before 2022 or did not include the ARASENS trial findings were tagged and captured within the PRISMA flowchart (Figure 1).
Study Selection
Abstracts were screened in a double-blind manner against predefined inclusion and exclusion criteria, with discrepancies resolved by a third reviewer. Full texts were reviewed to confirm their eligibility in a single-blind manner, and uncertainties were resolved by a senior reviewer. Patients with mHSPC met inclusion criteria if they had received docetaxel or ARATs (eg, AAP, enzalutamide, APA, or darolutamide) in any combination with ADT, radiation therapy, or standard of care for low-volume or developmental agents. Outcomes included OS, progression-free survival (PFS), adverse events (AEs), health-related quality of life (HRQOL), and other secondary end points. Eligible study designs included full publications and congress abstracts. Studies published in languages other than English were excluded.
To present the most up-to-date evidence, this review summarizes the meta-analyses (and ITCs), including the ARASENS trial results; publications not including the ARASENS trial are listed in Supplementary Table 2.
Data Extraction
Data were extracted into prespecified data-extraction tables by 1 reviewer and checked for quality by a second reviewer. Data were extracted based on methodology, outcomes (efficacy and AEs, including subgroup or secondary analyses), and treatment ranking (for ITCs only). Efficacy outcomes were extracted as hazard ratios (HRs) and safety outcomes as odds ratios, risk ratios, or HRs, with corresponding 95% CIs or credible intervals as reported in the publication.
Assessments from each ITC evaluating risk of bias (eg, Cochrane Risk of Bias 2) and rating the confidence and certainty of the results of a network meta-analysis were extracted, including the Grading of Recommendations Assessment, Development and Evaluation (GRADE)5 and Confidence in Network Meta-Analysis (CINeMA)6 approaches. Risk of bias and quality assessments were also extracted for each meta-analysis (without ITC).
Data Analysis
Data were summarized from meta-analyses (and ITCs) in tables and schematics, with a narrative summary. Credible intervals and CIs were assumed to be the same for ease of interpretation, and it was acknowledged that some studies used bayesian analysis, whereas others used frequentist statistical frameworks. This review of meta-analyses (and ITCs) compares included trials, treatment regimens, statistical methods, network inconsistency and heterogeneity tests, adjustment for trial-level factors, and estimated effect sizes and precision, as reported by each publication. A benefit was defined as an HR less than 1, and imprecision was described separately (where the 95% CI crossed the line of no effect).
Results
Included Study Population and Characteristics
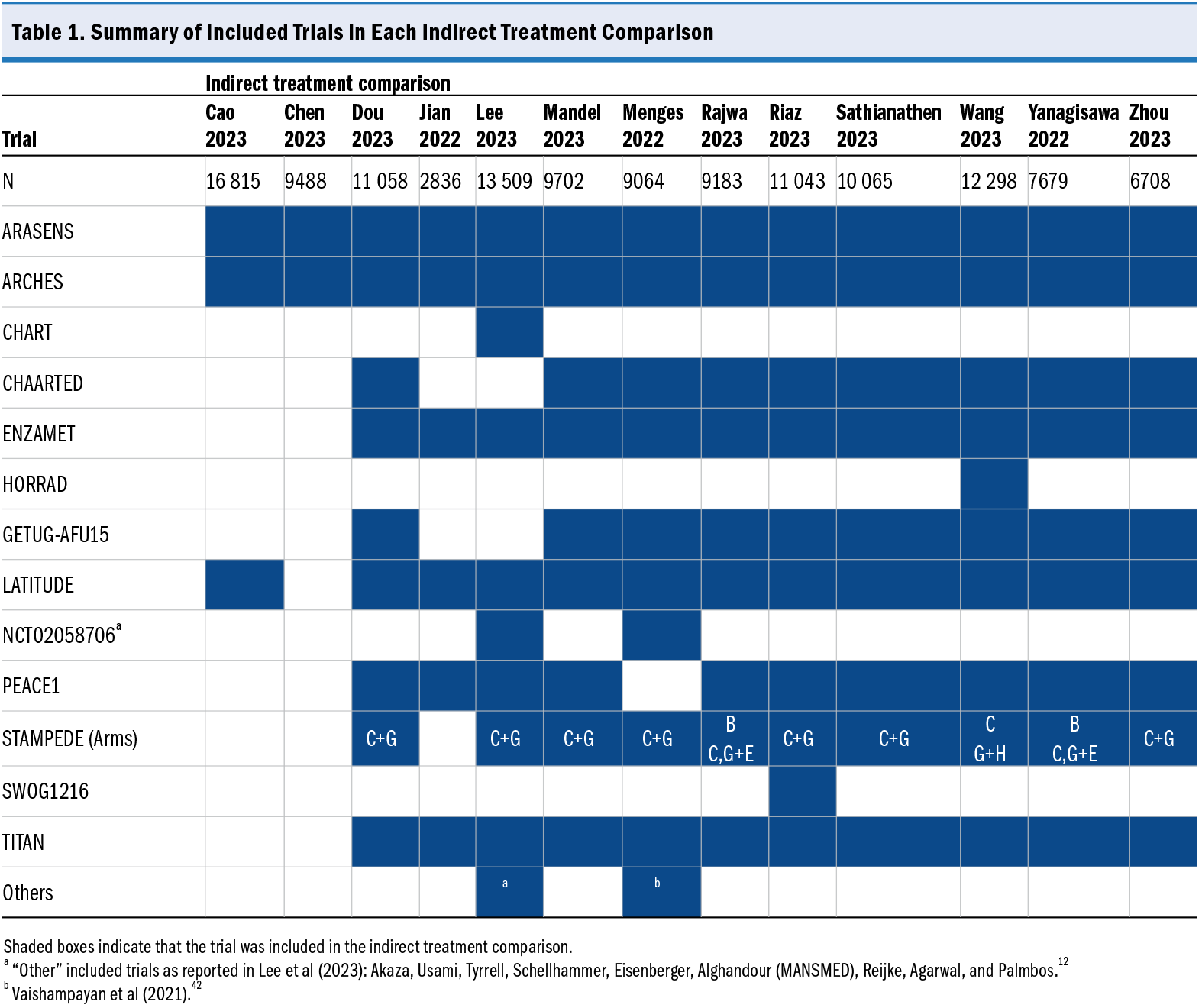
Thirty-one articles published through February 12, 2024, were included, as shown in the PRISMA flowchart (Figure 1). The identified publications included 13 ITCs (20 publications; Supplementary Table 3) of specific treatments (eg, darolutamide triple therapy vs docetaxel + ADT), 2 ITCs (3 publications) of treatment classes (eg, pooled ARAT triple therapy vs ARAT plus ADT or docetaxel plus ADT), and 8 meta-analyses (direct pairwise comparisons) of treatments (eg, ARAT triple therapy vs docetaxel plus ADT).
Methodology of Included ITCs and Meta-Analyses
Among the ITCs, there was variation in the number of trials included in each analysis (range, 2-17) (Table 1) and in statistical methods used (frequentist or bayesian network meta-analyses, random or fixed effects). Few ITCs reported the justification of the analysis model, with only Mandel et al7 and the living network meta-analysis from Riaz et al8 providing a rationale. Two ITCs described consideration of treatment effect modifiers,9,10 and few detailed tests were used to assess network inconsistency.10,11 Menges et al10 reported having assessed inconsistency according to epidemiologic criteria and the presence of potential effect modifiers; however, no further details are given. Wang et al11 described the assessment of inconsistency via a node-splitting model
(P < .05 was considered statistically significant). A minority of ITCs performed quality and certainty assessments for network outcomes (eg, GRADE or CINeMA).8-11 There were also differences across the identified meta-analyses, with variation in the number of trials per analysis (range, 5-10) and differences in the analytical models used. No ITCs considered variation in HRs over time using, for example, fractional polynomial models.
The majority of studies focused on the analysis of OS, with PFS (including the reconstituted or proxy end point ARASENS trial data) reported by 3 ITCs12-14 and 3 meta-analyses.15-17 Adverse events were reported in 8 ITCs and 4 meta-analyses. No ITCs or meta-analyses reported HRQOL outcomes.
Treatment vs Treatment Comparison
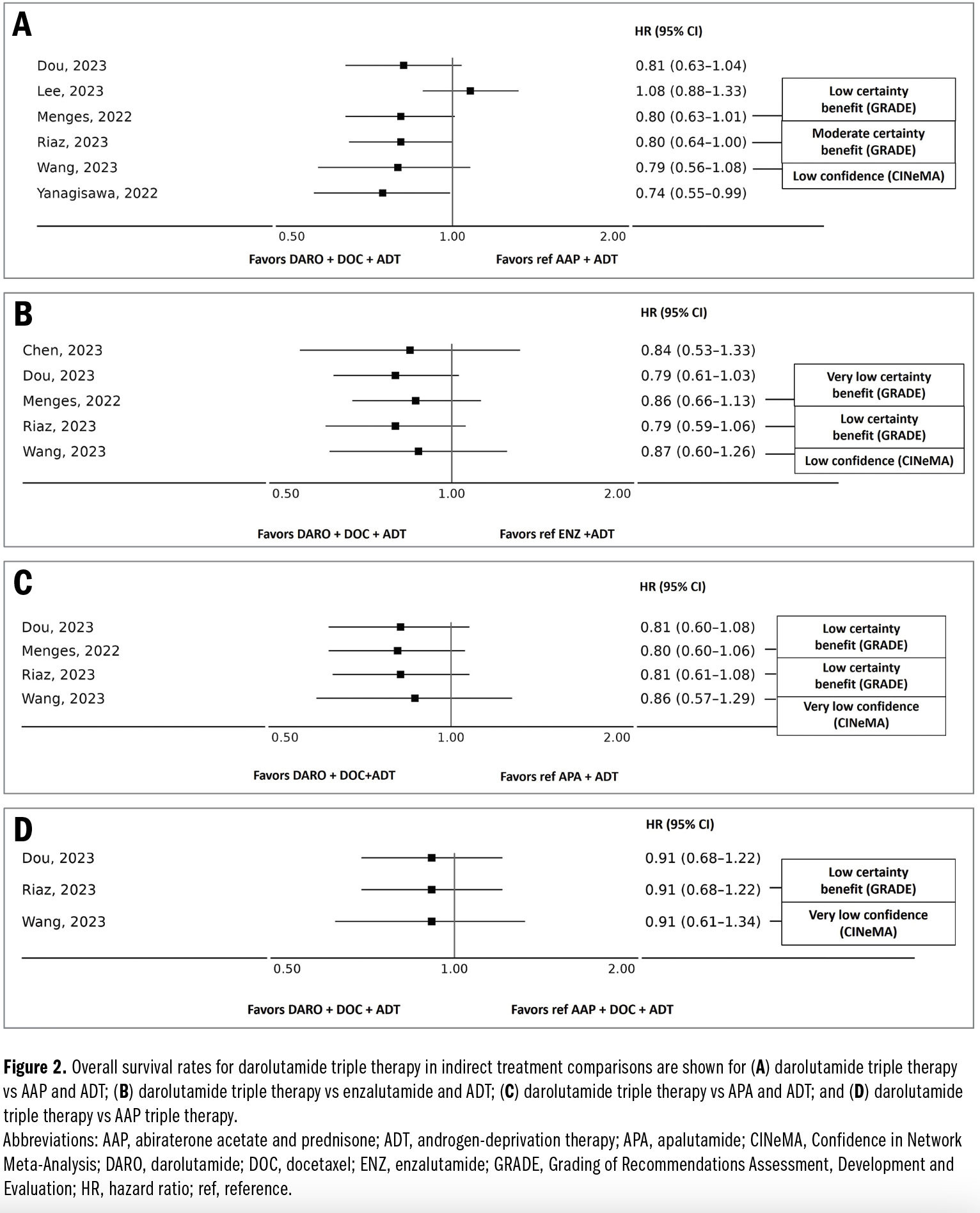
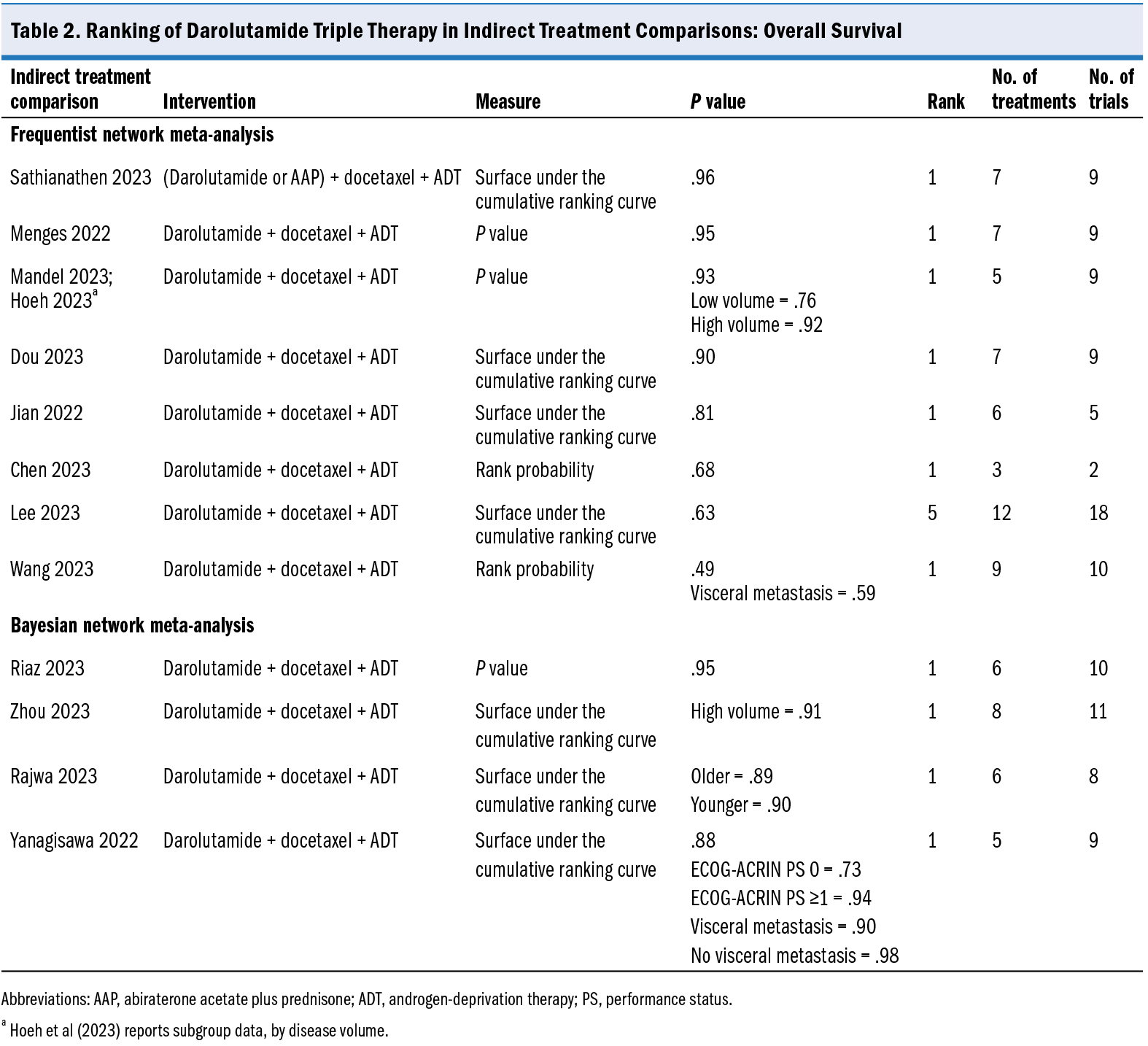
Indirect treatment comparisons consistently showed a significant benefit in OS with darolutamide triple therapy compared with ADT (4 ITCs) and docetaxel plus ADT (8 ITCs in line with ARASENS trial results; Supplementary Table 4). Darolutamide triple therapy demonstrated a benefit in OS compared with ARAT doublet therapies (AAP plus ADT, enzalutamide plus ADT, or APA plus ADT), but the confidence in this effect estimate is uncertain owing to imprecision (wide CIs) (Figure 2A-2C). Three ITCs reported the comparison of darolutamide triple therapy vs AAP triple therapy. The ITCs reported an HR that indicated a trend toward reduced risk of death with darolutamide triple therapy; however, the CI includes 1 (HR, 0.91 [95% CI, 0.68-1.22] [2 ITCs] and HR, 0.91 [95% CI, 0.61-1.34] [1 ITC]) with low certainty (GRADE) or very low confidence (CINeMA) (Figure 2D). Darolutamide triple therapy was consistently ranked highest for OS in 11 of 12 ITCs that reported ranking analyses, above ARAT doublet and triple therapies, except for Lee et al.12 Sathianathen et al14 included darolutamide or AAP triple therapy as a pooled treatment in their ranking analysis, which was ranked highest for OS. See Table 2 for a summary of the ranking of darolutamide triple therapy in each ITC.
One ITC included time to castration-resistant prostate cancer within their PFS analysis network, as evaluated in the ARASENS trial. The ITC reported darolutamide triple therapy compared with docetaxel plus ADT for this end point (HR, 0.47 [95% CI, 0.20-1.10]).14 Other ITCs included PFS but without ARASENS data owing to inconsistency in end point definitions among studies.
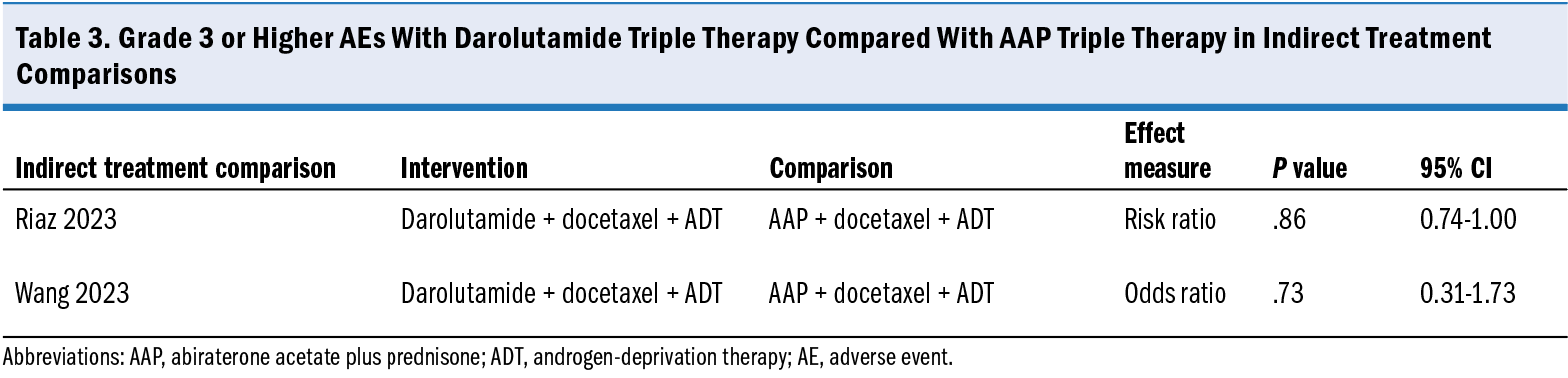
Of the ITCs that included ARASENS trial data, 8 studies reported AE analyses. The risk of grade 3 or higher AEs was greater with darolutamide triple therapy than with ARAT doublet therapies across 6 ITCs (GRADE, low to moderate certainty of evidence; CINeMA, very low confidence; Supplementary Table 5). There was a similar risk of grade 3 or higher AEs with darolutamide triple therapy and with docetaxel plus ADT across 4 ITCs, but the confidence in this result was rated as having low to moderate certainty with GRADE and very low confidence with CINeMA. Indirect treatment comparisons of AEs across triple therapies showed a lower risk of grade 3 or higher AEs for darolutamide triple therapy than for AAP triple therapy across 2 ITCs (GRADE, low certainty; CINeMA, very low confidence) (Table 3).8,11 Four ITCs ranked darolutamide triple therapy as the third to seventh treatment, and it was consistently ranked higher than AAP plus docetaxel plus ADT with respect to the incidence of grade 3 or higher AEs.8,11,18,19 Androgen-deprivation therapy alone was consistently ranked first, showing a favorable safety profile.8,11,18,19
Treatment Class vs Treatment Class Comparison in an ITC
Two ITCs across 3 publications reported OS for pooled ARAT triple therapies (including data from the ARASENS trial) compared with pooled ARAT doublet therapies. Naqvi et al20 showed increased OS with ARAT plus docetaxel plus ADT compared with docetaxel plus ADT (HR, 0.74 [95% CI, 0.66-0.84]).20 Androgen receptor axis–targeted therapy plus ADT in 2 ITCs21,22 reported an HR less than 1.0 for OS (Naqvi et al, HR, 0.97 [95% CI, 0.78-1.20]; Roy et al, HR, 0.89 [95% CI, 0.68-1.16]), but wide CIs indicated no evidence of a difference.21,22 Progression-free survival and AE outcomes were not reported for these 2 pooled analyses (ARASENS not included in networks).
Treatment Comparisons (Direct Pairwise Meta-Analysis Only)
Eight meta-analyses were identified, 4 of which reported OS for ARAT plus docetaxel plus ADT vs docetaxel plus ADT. Two of these meta-analyses reported OS for ARAT plus ADT with or without docetaxel vs ADT with or without docetaxel (Supplementary Table 6). The meta-analyses showed a consistent benefit in OS with ARAT plus docetaxel plus ADT compared with docetaxel plus ADT. Triple therapy compared with ADT alone was not reported by any of the included meta-analyses, with some noting that this comparison was not part of their objective and others noting that ADT alone does not represent current standard of care.15,23 Progression-free survival was reported in 3 meta-analyses, with a consistent survival benefit seen with ARAT plus docetaxel plus ADT vs docetaxel plus ADT (HR, 0.41-0.43) (Supplementary Table 6).15-17 Adverse events were reported in 4 meta-analyses of ARAT triple therapies, of which 3 reported grade 3 or higher AEs and 1 reported cardiovascular events, all favoring docetaxel plus ADT (Supplementary Table 6).16,17,24,25
Population Subgroups
Several ITCs and meta-analyses included subgroup analyses or secondary stratified analyses in specific populations. A summary of subgroup analyses reported in ITCs for OS is given in Supplementary Figure 1, including by disease volume (low or high), age (older [≥65 or ≥70 years] or younger patients [≤65 or ≤70 years]), presence of visceral metastasis, ECOG-ACRIN performance status (PS) (0 or ≥1), Gleason score (≥8 or <8), and recurrent (metachronous) or de novo (synchronous) disease. Notably, Hoeh et al26 reported a benefit in OS with ARAT triple therapy compared with ARAT plus ADT in patients with high-volume disease (Supplementary Figure 1A), with darolutamide triple therapy ranking first (P = .92), followed by AAP triple therapy
(P = .85). The OS estimates for patients with low-volume disease are imprecise, and there is therefore uncertainty around these results.26,27 Many subgroup analyses showed imprecise results, but there is some evidence to suggest larger benefits in OS with darolutamide triple therapy in younger patients than in older patients, and in patients with an ECOG-ACRIN PS of at least 1 than in patients with a PS of 0 (Supplementary Figure 1B and 1D). In addition, darolutamide triple therapy vs both ADT and docetaxel plus ADT demonstrated a significant benefit in OS in patients with de novo disease than in patients with recurrent disease (Supplementary Figure 1F).8,10,11,28 Additional subgroup analyses in publications reporting meta-analyses (without ITCs) are shown in Supplementary Table 6.
Zhou et al13 reported the time to castration-resistant prostate cancer end point as evaluated in ARASENS, within their PFS analysis network in patients with high-volume disease (HR, 0.41 [95% CI, 0.34-0.49]). No subgroup analyses were reported for AEs.
Discussion
We systematically identified ITCs and meta-analyses of darolutamide therapies in mHSPC that included the ARASENS trial, identifying 15 ITCs and 8 meta-analyses published between October 2022 and February 2024.
Indirect treatment comparisons consistently ranked darolutamide triple therapy highest among studied treatment options, including AAP doublet and triple therapies, in terms of OS benefit but with uncertainty, as indicated by wide CIs. Evidence for PFS was sparse; it was not analyzed in most ITCs because of differences in PFS definitions across trials. We note that the ARASENS trial measured castration-resistant prostate cancer–free survival, whereas other trials included clinical PFS or radiographic PFS, which rendered a comparison across these trials and end points inappropriate, as discussed in several meta-analytical reports. In terms of safety, ITCs of darolutamide triple therapy reported a lower rate of grade 3 or higher AEs compared with AAP triple therapy and reported a similar safety profile to docetaxel plus ADT. Compared with ARAT doublet therapy, the risk of grade 3 or higher AEs was higher (although imprecise) with darolutamide triple therapy. This risk is reflected in the ranking analysis, which ranked darolutamide triple therapy as the third to seventh treatment across ITCs (consistently higher than AAP triple therapy).
Two ITCs compared treatment classes and included ARASENS data. They both showed longer OS with ARAT plus docetaxel plus ADT (triple therapy) than with docetaxel plus ADT and had an OS benefit compared with ARAT plus ADT. Neither ITC reported PFS or AE outcomes that included ARASENS in its networks.
In meta-analyses (without ITCs), a consistent benefit in OS was demonstrated with ARAT triple therapy compared with docetaxel plus ADT (6 studies). The benefit in OS reported with triple therapies across ITCs in patients with mHSPC is consistent with clinical guidelines, including the National Comprehensive Cancer Network (v3.2024) (for patients with high-volume or metachronous metastases and for patients with low-volume or high-volume synchronous metastases)29 and the European Association of Urology,30 which recommend darolutamide or AAP triple therapy.
Subgroup analysis showed a benefit in OS for patients with high-volume disease with ARAT triple therapy compared with doublet therapy. Results for low-volume disease should be interpreted with caution given the lower numbers of patients in the analysis and the low event rates; triple therapy may still be a valid treatment option given this uncertainty. Secondary analyses suggested that being younger (compared with being older), having an ECOG-ACRIN PS of at least 1 (compared with having a PS of 0), and having de novo disease (compared with having recurrent disease) are prognostic of improved survival, although several analyses included imprecise effect estimates.
Although the findings of the meta-analyses (including ITCs) were generally consistent, there were methodological differences. One such difference was the number of trials included in each ITC, in part because of the objective of each study. For example, Chen et al31 focused on newer-generation ARATs and so included only second-generation ARATs (enzalutamide, APA, darolutamide) or placebo trials, whereas most other studies captured the efficacy and safety of all included trials for all available treatment options.
Lee et al32 reported the only ITC that showed a lower ranking of triple therapies over doublet therapies for OS. They used the intention-to-treat analysis for ARASENS unlike the other included ITCs. In addition, rezvilutamide was included from the CHART trial, which included a subpopulation of Asian men with de novo, high-volume mHSPC.32 Rezvilutamide has limited availability in the United States and the European Union, which may explain why CHART was not included in other ITCs. The ITC reported by Zhou et al13 was the only other ITC that included the CHART trial. They reported that in patients with high-volume disease, darolutamide triple therapy ranked the highest and as the best-performing regimen in terms of OS, but they did not report ITCs of triple therapies with doublet therapies.13
Analytic methods varied across included studies, which complicated comparisons. Most identified ITCs did not assess inconsistencies and assumed that there were few or no treatment effect modifiers (or population heterogeneity) without formally checking or documenting these considerations. Only 1 ITC described the assessment of inconsistency and used a node-splitting model with closed-loop comparisons all suggesting P > .111; however, it is uncertain whether the consistency assumptions made were justified across other ITCs because there is limited information reported. Another element of uncertainty is the use of HRs that are constant over time (proportional hazard assumption) despite trials having different follow-up durations and treatments potentially having different speeds of action. In addition, the choice of the analysis model for safety end points varied; ITCs calculated either odds ratios or risk ratios, which gave wider or narrower CIs and therefore different interpretations of results across conclusions. Most of the included ITCs in this review, with the exception of the ITC reported by Menges et al,10 did not account for the unique structure of the Systemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation of Drug Efficacy (STAMPEDE) study (ClinicalTrials.gov identifier NCT00268476), which was a multiarm, multistage, adaptive trial.33 Vale et al33 were the first to make this adjustment, which was later incorporated into individual patient data (IPD) analysis.34 In addition, most ITCs did not explore subgroup or sensitivity analyses other than for disease volume. More recent publications, however, are providing more of these types of analyses as updates or associated studies (eg, subanalyses according to ECOG-ACRIN PS and in patients with visceral metastasis).35,36 There was also an absence of population-adjusted ITCs,37 such as matching-adjusted indirect comparisons, multilevel network meta-regressions, and network meta-interpolations.38-40 In addition, there were differences in the trials themselves; for example, 2 trials had an open-label study design (Enzalutamide in First Line Androgen Deprivation Therapy for Metastatic Prostate Cancer [ENZAMET; ClinicalTrials.gov identifier NCT02446405] and A Phase III Study for Patients With Metastatic Hormone-naïve Prostate Cancer [PEACE1; ClinicalTrials.gov identifier NCT01957436]).
There was a lack of HRQOL outcomes identified from ITCs of darolutamide, in part owing to ARASENS using the National Comprehensive Cancer Network/Functional Assessment of Cancer Therapy Prostate Cancer Symptom Index 17-item version instrument, which was not used across other trials, meaning that no comparisons were possible. Health-related QOL outcomes were assessed in a minority of ITCs, despite being measured across trials. Menges et al8 and Riaz et al10 reported that 8 and 7 of 10 trials reported data on HRQOL, respectively. Menges et al8 described a short-term (3-month to 6-month) decrease in HRQOL with ADT plus docetaxel and reported a potential benefit of ADT plus AAP in HRQOL at up to 24 months of follow-up compared with ADT alone. Riaz et al10 noted that evidence is still emerging. If consistent patient-reported outcome instruments are used in future trials, comparisons of HRQOL will be possible.
This review’s findings are consistent with those of Fisher et al,4 who also examined results and methods across published ITCs in mHSPC and described variation in eligibility criteria and statistical methodology between identified ITCs. Despite this variation, the findings of Fisher et al4 are in agreement with the similarity in results reported across ITCs in this review. Our review has broader inclusion criteria for analysis type (including meta-analyses as well as ITCs), has a unique objective in comparing analyses of darolutamide in the published literature, and is more up-to-date owing to our later search cutoff date.
Company-sponsored studies are rarely included in IPD network meta-analyses, and we note that no IPD analyses that included ARASENS data were identified in the literature. An IPD of the GETUG-AFU15, Androgen Ablation Therapy With or Without Chemotherapy in Treating Patients With Metastatic Prostate Cancer (CHAARTED; ClinicalTrials.gov identifier NCT00309985), and STAMPEDE trials has been published, describing the benefit in OS of adding docetaxel to ADT, including in treatment of patients with high-volume disease.34 We also note the publication of the Darolutamide in Addition to ADT Versus ADT in Metastatic Hormone-sensitive Prostate Cancer (ARANOTE; ClinicalTrials.gov identifier NCT04736199) phase 3 randomized controlled trial, which reports that darolutamide plus ADT significantly improved radiographic PFS, reducing the risk of progression or death by 46% compared with placebo plus ADT, with consistent benefits across subgroups, including high-volume and low-volume disease.41 Adverse events were similar between the darolutamide and placebo groups. This review followed a robust process to minimize the limitations of systematic reviews, with an extensive search strategy, and a protocol registered a priori in PROSPERO. We acknowledge, however, that a limitation of this review is the requirement for analyses to include ARASENS.
Conclusions
Despite methodologic variability and imprecision in results, ITCs consistently ranked darolutamide triple therapy highly for OS over other treatment options. Consistent results from ITCs of greater OS with darolutamide triple therapy compared with docetaxel plus ADT were in line with evidence from ARASENS.3 There remains a need to standardize ITC methodology for PFS and safety end points to ensure robustness and interpretability of findings and to optimize treatment decision-making.
References
1. Nubeqa. Prescribing information. Bayer HealthCare Pharmaceuticals, Inc; 2022. Accessed March 6, 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/212099s002lbl.pdf
2. European Medicines Agency. Nubeqa (darolutamide). 2023. Accessed March 6, 2024. https://www.ema.europa.eu/en/documents/overview/nubeqa-epar-medicine-overview_en.pdf
3. Smith MR, Hussain M, Saad F, et al; ARASENS Trial Investigators. Darolutamide and survival in metastatic, hormone-sensitive prostate cancer. N Engl J Med. 2022;386(12):1132-1142. doi:10.1056/NEJMoa2119115
4. Fisher DJ, Burdett S, Vale C, White IR, Tierney JF. Duplicated network meta-analysis in advanced prostate cancer: a case study and recommendations for change. Syst Rev. 2022;11(1):274. doi:10.1186/s13643-022-02137-6
5. Chaimani A, Caldwell DM, Li T, Higgins JPT, Salanti G. Chapter 11: Undertaking network meta-analyses. In: Higgins J. Cochrane Handbook for Systematic Reviews of Interventions, version 6.4. Updated August 2023. Accessed March 11, 2024. https://training.cochrane.org/handbook/current/chapter-11#section-11-5
6. Nikolakopoulou A, Higgins JPT, Papakonstantinou T, et al. CINeMA: an approach for assessing confidence in the results of a network meta-analysis. PLoS Med. 2020;17(4):e1003082. doi:10.1371/journal.pmed.1003082
7. Mandel P, Hoeh B, Wenzel M, et al. Triplet or doublet therapy in metastatic hormone-sensitive prostate cancer patients: a systematic review and network meta-analysis. Eur Urol Focus. 2023;9(1):96-105. doi:10.1016/j.euf.2022.08.007
8. Riaz IB, Naqvi SAA, He H, et al. First-line systemic treatment options for metastatic castration-sensitive prostate cancer: a living systematic review and network meta-analysis. JAMA Oncol. 2023;9(5):635-645. doi:10.1001/jamaoncol.2022.7762
9. Jian T, Zhan Y, Hu K, et al. Systemic triplet therapy for metastatic hormone-sensitive prostate cancer: a systematic review and network meta-analysis. Front Pharmacol. 2022;13:955925. doi:10.3389/fphar.2022.955925
10. Menges D, Yebyo HG, Sivec-Muniz S, et al. Treatments for metastatic hormone-sensitive prostate cancer: systematic review, network meta-analysis, and benefit-harm assessment. Eur Urol Oncol. 2022;5(6):605-616. doi:10.1016/j.euo.2022.04.007
11. Wang L, Li C, Zhao Z, et al. Comparison of doublet and triplet therapies for metastatic hormone-sensitive prostate cancer: a systematic review and network meta-analysis. Front Oncol. 2023;13:1104242. doi:10.3389/fonc.2023.1104242
12. Lee YS, Kim SH, Tae JH, et al. Oral chemotherapeutic agents in metastatic hormone-sensitive prostate cancer: a network meta-analysis of randomized controlled trials. Prostate Int. 2023;11(3):159-166. doi:10.1016/j.prnil.2023.06.003
13. Zhou Z, Liu S, Mei J, Liu T, Liu F, Zhang G. Systemic therapies for high-volume metastatic hormone-sensitive prostate cancer: a network meta-analysis. Acta Oncol. 2023;62(9):1083-1090. doi:10.1080/0284186X.2023.2241985
14. Sathianathen NJ, Pan HYC, Lawrentschuk N, et al. Emergence of triplet therapy for metastatic castration-sensitive prostate cancer: an updated systematic review and network meta-analysis. Urol Oncol. 2023;41(5):233-239. doi:10.1016/j.urolonc.2022.10.016
15. Fallara G, Robesti D, Nocera L, et al. Chemotherapy and advanced androgen blockage, alone or combined, for metastatic hormone-sensitive prostate cancer a systematic review and meta-analysis. Cancer Treat Rev. 2022;110:102441. doi:10.1016/j.ctrv.2022.102441
16. Fiorica F, Buttigliero C, Grigolato D, et al. Addition of new androgen receptor pathway inhibitors to docetaxel and androgen deprivation therapy in metastatic hormone-sensitive prostate cancer: a systematic review and metanalysis. Curr Oncol. 2022;29(12):9511-9524. doi:10.3390/curroncol29120747
17. Ramos-Esquivel A, Garita-Rojas E, Masis-Marroquín A. A systematic review and meta-analysis on overall survival, failure-free survival and safety outcomes in patients with metastatic hormone-sensitive prostate cancer treated with new anti-androgens. Anticancer Drugs. 2023;34(3):405-412. doi:10.1097/CAD.0000000000001419
18. Cao B, Kim M, Reizine NM, Moreira DM. Adverse events and androgen receptor signaling inhibitors in the treatment of prostate cancer: a systematic review and multivariate network meta-analysis. Eur Urol Oncol. 2023;6(3):237-250. doi:10.1016/j.euo.2023.01.001
19. Dou M, Liang H, Liu Y, et al. Based on ARASENS trial: efficacy and safety of darolutamide as an emerging option of endocrinotherapy for metastatic hormone-sensitive prostate cancer—an updated systematic review and network meta-analysis. J Cancer Res Clin Oncol. 2023;149(10):7017-7027. doi:10.1007/s00432-023-04658-6
20. Naqvi SAA, Riaz ZB, Riaz A, et al. Indirect comparisons of triplet therapy as compared to novel hormonal therapy doublets in patients with metastatic castration sensitive prostate cancer. J Clin Oncol. 2022;40(16_suppl):5083. doi:10.1200/JCO.2022.40.16_suppl.5083
21. Naqvi SAA, Riaz ZB, Riaz A, et al. Triplet therapy in metastatic castration-sensitive prostate cancer: a systematic review and meta-analysis. J Clin Oncol. 2022;40(6 suppl):136. doi:10.1200/JCO.2022.40.6_suppl.136
22. Roy S, Sayyid R, Saad F, et al. Addition of docetaxel to androgen receptor axis-targeted therapy and androgen deprivation therapy in metastatic hormone-sensitive prostate cancer: a network meta-analysis. Eur Urol Oncol. 2022;5(5):494-502. doi:10.1016/j.euo.2022.06.003
23. Ciccarese C, Iacovelli R, Sternberg CN, Gillessen S, Tortora G, Fizazi K. Triplet therapy with androgen deprivation, docetaxel, and androgen receptor signalling inhibitors in metastatic castration-sensitive prostate cancer: a meta-analysis. Eur J Cancer. 2022;173:276-284. doi:10.1016/j.ejca.2022.07.011
24. Maiorano BA, De Giorgi U, Roviello G, et al. Addition of androgen receptor-targeted agents to androgen-deprivation therapy and docetaxel in metastatic hormone-sensitive prostate cancer: a systematic review and meta-analysis. ESMO Open. 2022;7(5):100575. doi:10.1016/j.esmoop.2022.100575
25. El-Taji O, Jones C, Brown MD, Clarke NW, Sachdeva A. 1820P Cardiovascular effects of androgen receptor signalling inhibitors in the treatment of advanced and metastatic prostate cancer: a systematic review and meta-analysis of randomised controlled trials. Ann Oncol. 2023;34(suppl 2):S986. doi:10.1016/j.annonc.2023.09.2768
26. Hoeh B, Garcia CC, Wenzel M, et al. Triplet or doublet therapy in metastatic hormone-sensitive prostate cancer: updated network meta-analysis stratified by disease volume. Eur Urol Focus. 2023;9(5):838-842. doi:10.1016/j.euf.2023.03.024
27. Naqvi SAA, Riaz IB, He H, et al. The role of volume of disease for treatment selection in patients with metastatic castration sensitive prostate cancer (mCSPC): a living meta-analysis. J Clin Oncol. 2023;41(16_suppl):5088. doi:10.1200/JCO.2023.41.16_suppl.5088
28. Yanagisawa T, Rajwa P, Thibault C, et al. Androgen receptor signaling inhibitors in addition to docetaxel with androgen deprivation therapy for metastatic hormone-sensitive prostate cancer: a systematic review and meta-analysis. Eur Urol. 2022;82(6):584-598. doi:10.1016/j.eururo.2022.08.002
29. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Prostate Cancer Version 1.2025. Updated December 4, 2024. Accessed February 21, 2025. https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf
30. EAU Guidelines. Presented at: EAU Annual Congress; 2024; Paris, France. Accessed March 12, 2025. https://uroweb.org/guidelines/prostate-cancer
31. Chen X, Wang Q, Pan Y, et al. Comparative efficacy of second-generation androgen receptor inhibitors for treating prostate cancer: a systematic review and network meta-analysis. Front Endocrinol (Lausanne). 2023;14:1134719. doi:10.3389/fendo.2023.1134719
32. Singh K. Rezvilutamide for metastatic castration-sensitive prostate cancer: CHART trial. Indian J Urol. 2023;39(2):171-172. doi:10.4103/iju.iju_336_22
33. Vale CL, Fisher DJ, White IR, et al. What is the optimal systemic treatment of men with metastatic, hormone-naive prostate cancer? A STOPCAP systematic review and network meta-analysis. Ann Oncol. 2018;29(5):1249-1257. doi:10.1093/annonc/mdy071
34. Vale CL, Fisher DJ, Godolphin PJ, et al; STOPCAP Collaboration. Which patients with metastatic hormone-sensitive prostate cancer benefit from docetaxel: a systematic review and meta-analysis of individual participant data from randomised trials. Lancet Oncol. 2023;24(7):783-797. doi:10.1016/s1470-2045(23)00230-9
35. Fukuokaya W, Mori K, Yanagisawa T, Akazawa K, Shimomura T, Kimura T. Association between concomitant proton pump inhibitor use and survival of patients with metastatic prostate cancer receiving abiraterone acetate: a post-hoc analysis of pooled data from three randomized controlled trials. Prostate Cancer Prostatic Dis. 2024;27(3):444-450. doi:10.1038/s41391-023-00695-x
36. Yanagisawa T, Kawada T, Mori K, et al. Impact of performance status on efficacy of systemic therapy for prostate cancer: a meta-analysis. BJU Int. 2023;132(4):365-379. doi:10.1111/bju.16106
37. Phillippo DM, Ades AE, Dias S, Palmer S, Abrams KR, Welton NJ. Methods for population-adjusted indirect comparisons in health technology appraisal. Med Decis Making. 2018;38(2):200-211. doi:10.1177/0272989X17725740
38. Harari O, Soltanifar M, Cappelleri JC, et al. Network meta-interpolation: effect modification adjustment in network meta-analysis using subgroup analyses. Res Synth Methods. 2023;14(2):211-233. doi:10.1002/jrsm.1608
39. Phillippo DM, Dias S, Ades AE, et al. Multilevel network meta-regression for population-adjusted treatment comparisons. J R Stat Soc Ser A Stat Soc. 2020;183(3):1189-1210. doi:10.1111/rssa.12579
40. Signorovitch JE, Sikirica V, Erder MH, et al. Matching-adjusted indirect comparisons: a new tool for timely comparative effectiveness research. Value Health. 2012;15(6):940-947. doi:10.1016/j.jval.2012.05.004
41. Saad F, Vjaters E, Shore N, et al; ARANOTE Study Investigators. Darolutamide in combination with androgen-deprivation therapy in patients with metastatic hormone-sensitive prostate cancer from the phase III ARANOTE trial. J Clin Oncol. 2024;42(36):4271-4281. doi:10.1200/JCO-24-01798
42. Vaishampayan UN, Heilbrun LK, Monk P III, et al. Clinical efficacy of enzalutamide vs bicalutamide combined with androgen deprivation therapy in men with metastatic hormone-sensitive prostate cancer: a randomized clinical trial. JAMA Netw Open. 2021;4(1):e2034633. doi:10.1001/jamanetworkopen.2020.34633
Article Information
Published: 3/31/2025.
Conflict of Interest Disclosures: N.S. has received fees from Amgen, AstraZeneca, Bayer, Dendreon, Ferring Pharmaceuticals, Genentech, Janssen Scientific Affairs, Medivation/Astellas, Myovant Sciences, Pfizer, and Tolmar. A.K.M. has received fees from AAA Pharmaceutical, Astellas, AstraZeneca, Bayer, Exelixis, Lantheus, Merck, Myovant Sciences, Myriad, Novartis, Pfizer, Sanofi, and Telix. N.P., C.M., and S.C. are Bayer Pharmaceuticals employees and may own Bayer stock and/or options. S.H. and C.S.E. are employees of Oxford PharmaGenesis, who provide consulting services to Bayer. H.T. owns stock in Clifton Insight, which has received consulting fees from Amicus Therapeutics, Bayer, Baxter, Eisai, Janssen, Lundbeck, Merck, Novartis, Novo Nordisk, Pfizer, and Roche. M.B. has received fees from AAA Pharmaceutical, Astellas, AstraZeneca, Bayer, Bristol Myers Squibb, Eisai, Eli Lilly, EUSA Pharma, Exelixis, Gilead, Janssen, Merck, Merck Sharp & Dohme, Novartis, and Pfizer.
Funding/Support: This study was supported by Bayer.
Author Contributions: N.P., S.H., and C.S.E. had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. N.P. and N.S. conceived of and designed the study. N.P., S.H., and C.S.E. acquired the data. All authors analyzed and interpreted the data and drafted the manuscript. N.S., A.K.M., and H.T. performed critical revision of the manuscript for important intellectual content. S.H. provided administrative, technical, and material support. C.S.E. provided supervision.
Data Availability Statement: All data presented are in the public domain.
Supplementary Material: Supplementary material is available at Reviews in Urology online.